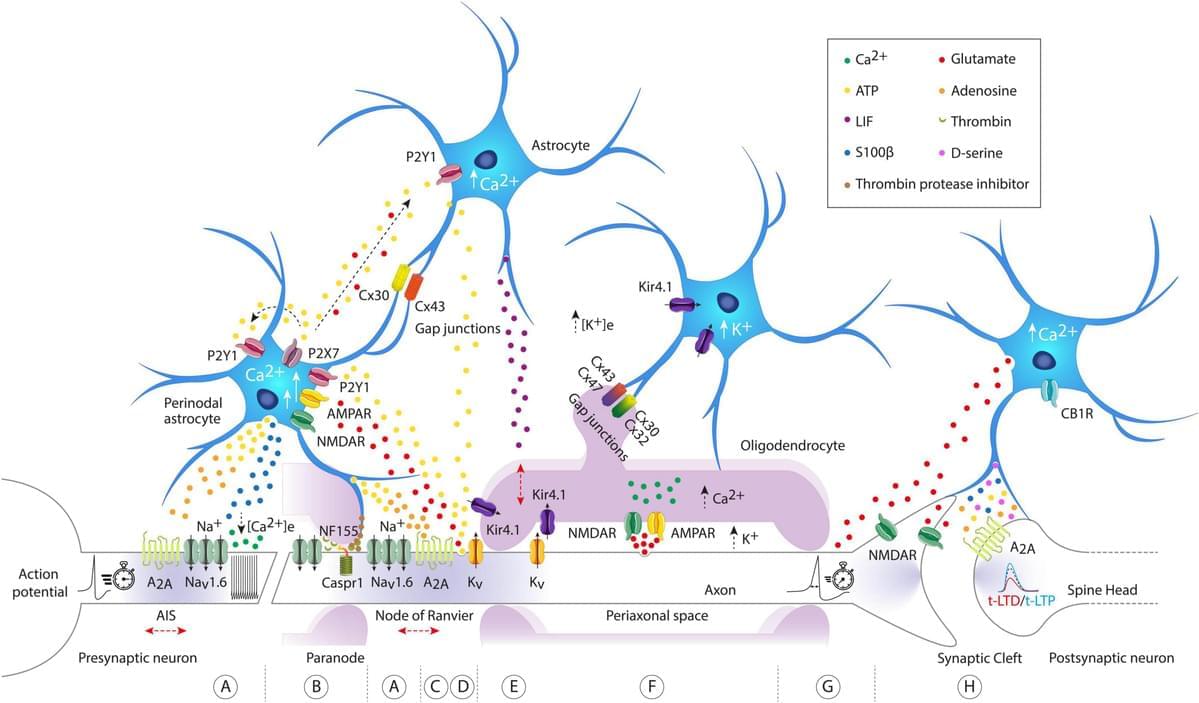
Neuronal network display various local or global mechanisms to allow information storage and transfer in the brain. From synaptic to intrinsic plasticity, the rules of input–output function modulation have been well characterized in neurons. In the past years, astrocytes have been suggested to increase the computational power of the brain and we are only just starting to uncover their role in information processing. Astrocytes maintain a close bidirectional communication with neurons to modify neuronal network excitability, transmission, axonal conduction, and plasticity through various mechanisms including the release of gliotransmitters or local ion homeostasis. Astrocytes have been significantly studied in the context of long-term or short-term synaptic plasticity, but this is not the only mechanism involved in memory formation. Plasticity of intrinsic neuronal excitability also participates in memory storage through regulation of voltage-gated ion channels or axonal morphological changes. Yet, the contribution of astrocytes to these other forms of non-synaptic plasticity remains to be investigated. In this review, we summarized the recent advances on the role of astrocytes in different forms of plasticity and discuss new directions and ideas to be explored regarding astrocytes-neuronal communication and regulation of plasticity.
The rules governing changes in synaptic and intrinsic plasticity are diverse and complex, sometimes synergistic and sometimes not (Debanne et al., 2019). Most studies have been neuro-centric, despite growing evidence that astrocytes can intervene or interact to modify or modulate synaptic transmission (Araque et al., 1998; Jourdain et al., 2007; Bonansco et al., 2011), input integration, neuronal excitability (Tan et al., 2017), spike waveform or axonal conductivity (Sasaki et al., 2011; Lezmy et al., 2021). Astrocytes can detect neuronal activity, and depending on the firing rate of action potentials (APs), they can not only release gliotransmitters such as adenosine or glutamate (Hamilton et al., 2008; Lezmy et al., 2021), but also trigger intracellular calcium ([Ca2+]i) oscillations at different frequencies (Pasti et al., 1997).