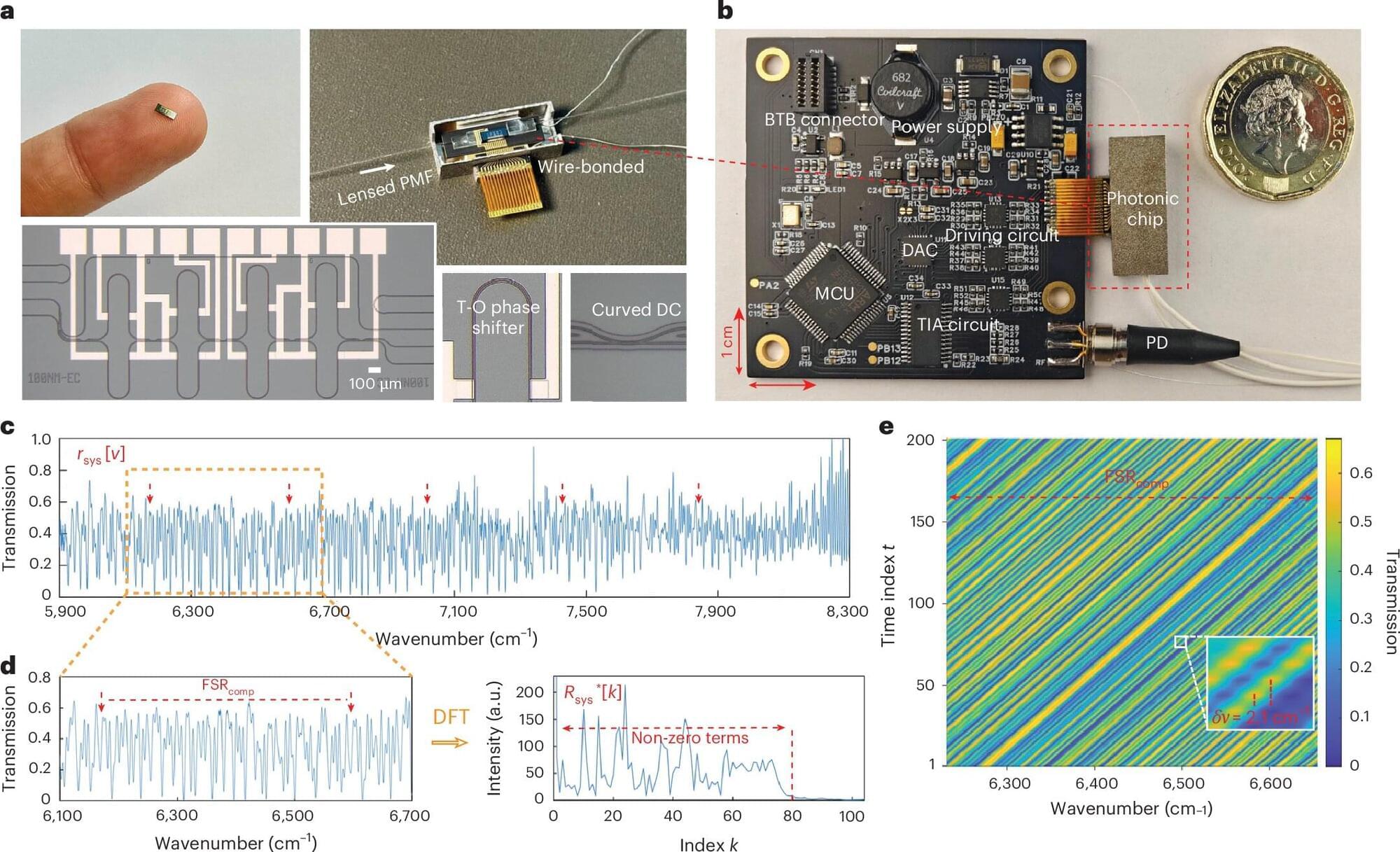
Researchers from the University of Cambridge and GlitterinTech, a startup founded by the same research group, have unveiled a fundamentally new type of optical spectrometer that delivers laboratory-grade precision in a device small enough to be embedded in portable and wearable technologies. By rethinking how spectra are measured and processed, the team has demonstrated a spectrometer costing only around $10, operating at a centimeter scale, and capable of applications ranging from industrial quality control to real-time health care monitoring.
Optical spectrometers underpin countless technologies, from chemical analysis and manufacturing to environmental sensing and medicine. Yet shrinking these instruments has historically involved painful trade-offs: Miniaturized devices typically sacrifice bandwidth, resolution or accuracy, limiting them to rough identification rather than true metrological measurements. The newly reported convolutional spectrometer overcomes these barriers by introducing a conceptually elegant operating principle grounded in the convolution theorem, offering unprecedented performance metrics compared with existing dispersive, Fourier-transform and reconstructive spectrometers.