Joe Betts-LaCroix and Retro Biosciences recently raised funding at a $1.8 billion valuation. In his first podcast appearance since the announcement, Joe shares his vision for extending healthy human lifespan and the breakthroughs driving the longevity industry forward.
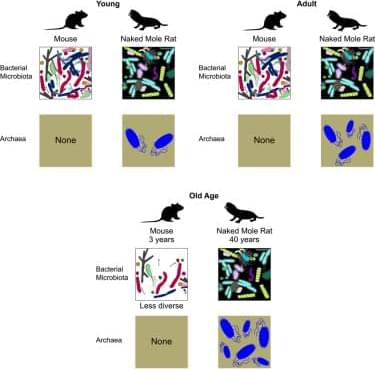
Joe Betts-LaCroix explains why aging is becoming a legitimate scientific target. He shares how new discoveries are turning longevity from speculation into measurable biology.
The conversation explores how AI is accelerating research, while highlighting why biology remains one of the hardest problems to solve. Even with smarter models, real-world testing and clinical trials still take time.
Joe also discusses Alzheimer’s, partial cellular reprogramming, and the future of longevity medicine. He shares why exercise remains the best longevity tool available today and what the next decade could look like for human health.
Joe is the CEO of Retro Biosciences and a longtime entrepreneur focused on science and technology. His mission is to extend healthy human lifespan and accelerate breakthroughs in aging research.
This episode is brought to you by NADclinic, the go-to destination for longevity and human performance. Check them out at https://nadclinic.com.