A new MIT-designed lidar chip uses specially engineered antennas to reduce interference and widen the sensor’s field of view.


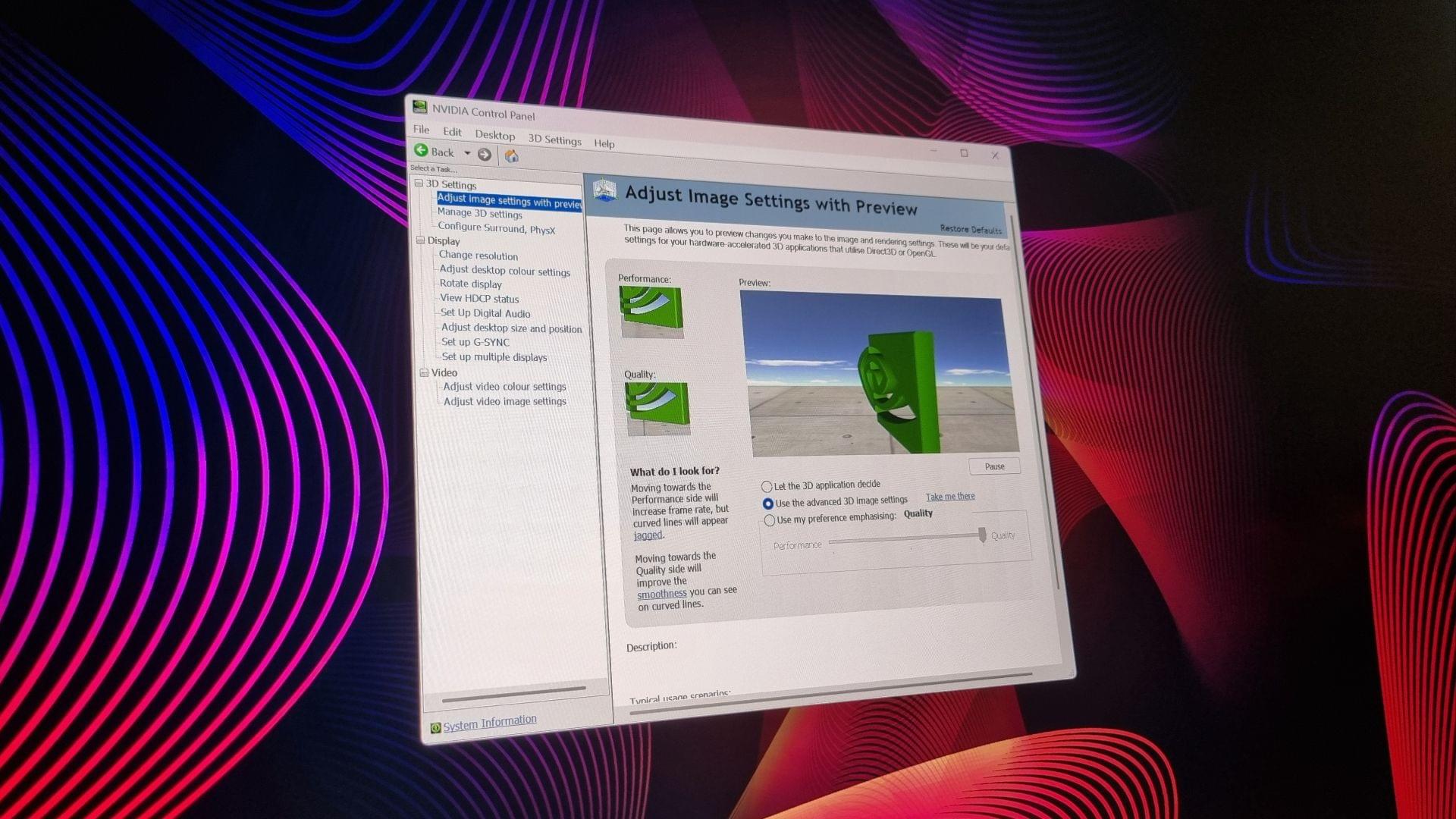
And two, the Nvidia App can be quite an online-focused affair in a way the old Control Panel was not. Besides the greyed-out checkbox in the Privacy settings labelled “required data” (something Nvidia explains is “Data that is necessary for Nvidia App to operate and cannot be switched off”), it can also lag quite badly on an unstable connection. In my personal experience, anyway.
Still, change comes for us all. I’ll miss the Control Panel’s classic rotating 3D image preview, the charmingly old-school HDCP menu that shows a rendering of an ancient Nvidia GPU plugged into what looks suspiciously like a plasma TV, and of course, the old Global Settings and Program Settings tabs with all of their many intricacies.
But is it progress? Perhaps. You can pry the Windows Control Panel from my cold, dead hands, though. That old clunker simply refuses to die, although I don’t think it’ll be that long before I write a similar obituary.

When we think about highly sensitive medical testing, we often imagine a hospital laboratory filled with large instruments, trained technicians, and carefully controlled conditions. This is especially true for optical biosensing, where scientists try to detect extremely small changes caused by biomolecules binding to a sensor surface.
These tiny changes can carry important information about disease, treatment response, or biological function. But detecting them often requires precise spectrometers, stable light sources, and carefully aligned instruments. This makes many advanced biosensing technologies powerful in the laboratory, but difficult to use in smaller clinics, remote regions, or point-of-care settings.
In our recent study, now published in Nature Photonics, we asked a simple question: Can we make high-performance label-free biosensing smaller, more robust, and easier to scale, without sacrificing sensitivity?


Engineers at Northwestern University have created printed artificial neurons that go beyond imitation and can directly interact with real brain cells. These flexible, low-cost devices produce electrical signals that closely resemble those generated by living neurons, allowing them to activate biological brain tissue.
In experiments using slices of mouse brain, the artificial neurons successfully triggered responses in real neurons. This result shows a new level of compatibility between electronic devices and living neural systems.
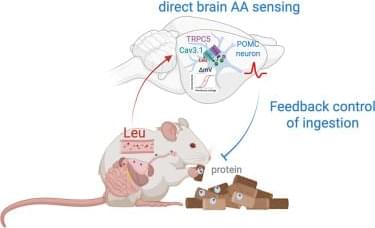
Online now: Tsang et al. identify the T-type calcium channel Cav3.1 as a neuronal leucine sensor in hypothalamic POMC neurons. Leucine directly binds Cav3.1, lowering its activation threshold. Loss of Cav3.1 in POMC neurons abolishes high-protein diet-induced appetite suppression, while pharmacological activation promotes weight loss and potentiates the effects of anorectic agents.

“It’s nearly invisible to the naked eye but our instruments give rise to a vision of swaths of scintillating corona glowing as thunderstorms pass overhead,” said Patrick McFarland. [ https://www.labroots.com/trending/earth-and-the-environment/…s-storms-2](https://www.labroots.com/trending/earth-and-the-environment/…s-storms-2)
Can storms cause electrical discharges on trees? This is something that has eluded scientists for decades, but a recent study published in Geophysical Research Letters might finally have an answer as a team of researchers collected first-time images of trees emanating ultraviolet (UV) light during a thunderstorm. This study has the potential to help scientists better understand how weather could affect plants worldwide.
For the study, the researchers traveled to Florida, which is known for its frequent and powerful thunderstorms, with the goal of imaging electric pulses that appear at the tips of trees during such storms. The primary motivation behind the study was to provide direct evidence of an atmospheric phenomenon that had been hypothesized since the 1930s but never captured in nature. During this time, scientists suspected that trees emit light during thunderstorms due to the electrical discharges but never had direct evidence.
Using a customized camera capable of capturing only corona light, lightning, and fire, this team of researchers successfully captured first-time evidence of trees emitting UV light during a Florida thunderstorm. These findings are crucial in helping researchers better understand the link between atmospheric electricity and forest ecology.
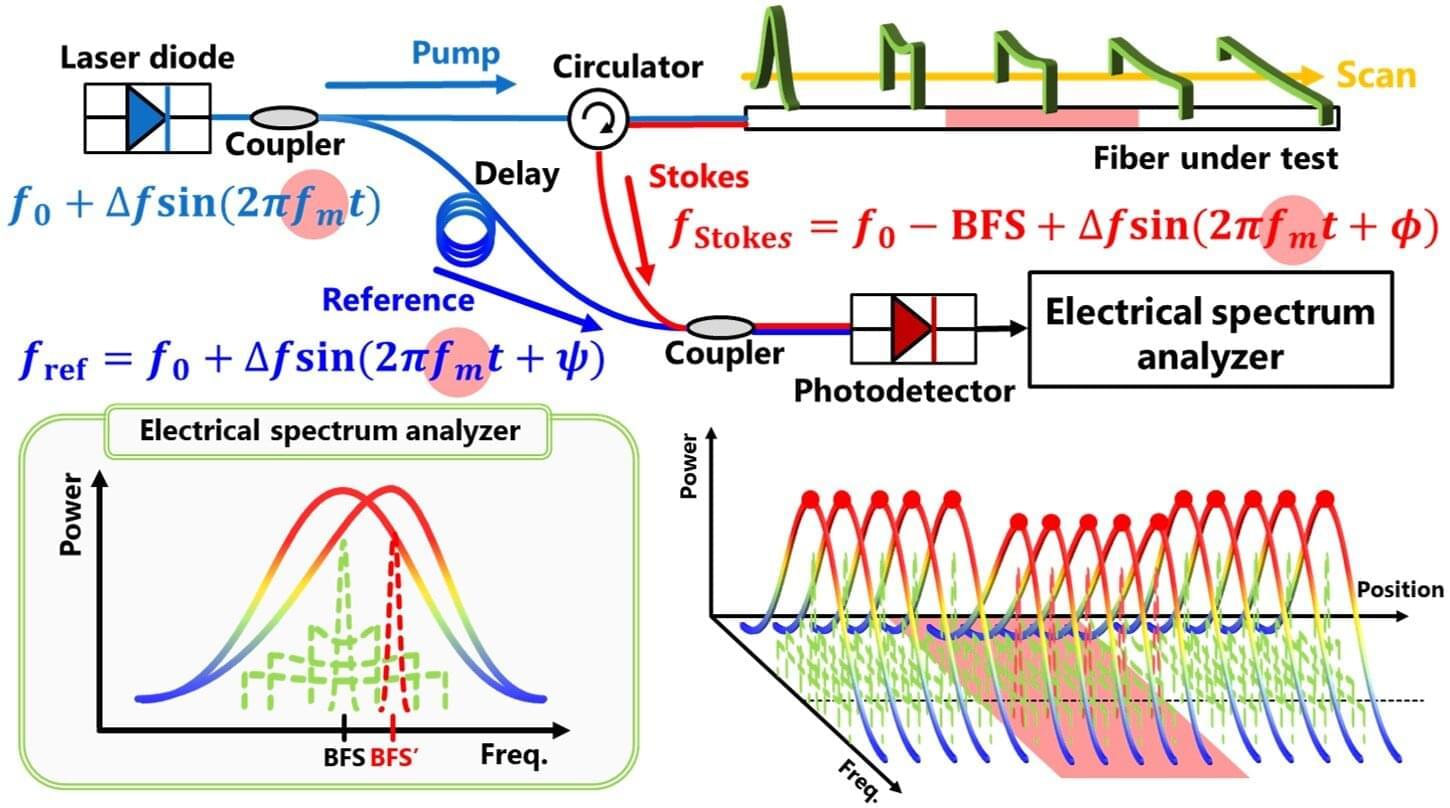
Distributed fiber-optic sensors are widely used to monitor temperature and strain in infrastructure, but their spatial resolution has long been limited. In a new study, researchers from Shibaura Institute of Technology and Yokohama National University, Japan, have demonstrated that operating near a previously avoided frequency regime and suppressing signal distortions allows reflection-based sensing to achieve a world-record spatial resolution of 6 mm among single-end-access configurations. This enables precise monitoring of temperature and strain in infrastructure.
Distributed fiber-optic sensing technologies play a crucial role in monitoring temperature and strain across large structures such as bridges, tunnels, pipelines, and buildings. Unlike conventional point sensors, distributed fiber-optic sensors provide continuous measurements along their entire length, allowing early detection of damage or abnormal conditions. However, one persistent challenge has been spatial resolution—the ability to pinpoint exactly where a change occurs. Improving resolution without complicating system design has remained a central goal in fiber-optic sensing research.
One promising technique, known as Brillouin optical correlation-domain reflectometry (BOCDR), enables distributed sensing using light injected from only one end of the fiber. This reflection-based configuration simplifies installation and allows measurements even if the fiber is damaged. BOCDR also offers higher spatial resolution than many other Brillouin-based methods. Yet, its performance has been constrained by a widely accepted assumption: operating near or beyond the Brillouin bandwidth, a frequency range intrinsic to the fiber, was believed to cause unstable signals and unreliable measurements. As a result, this operating regime has largely been avoided, limiting achievable resolution.

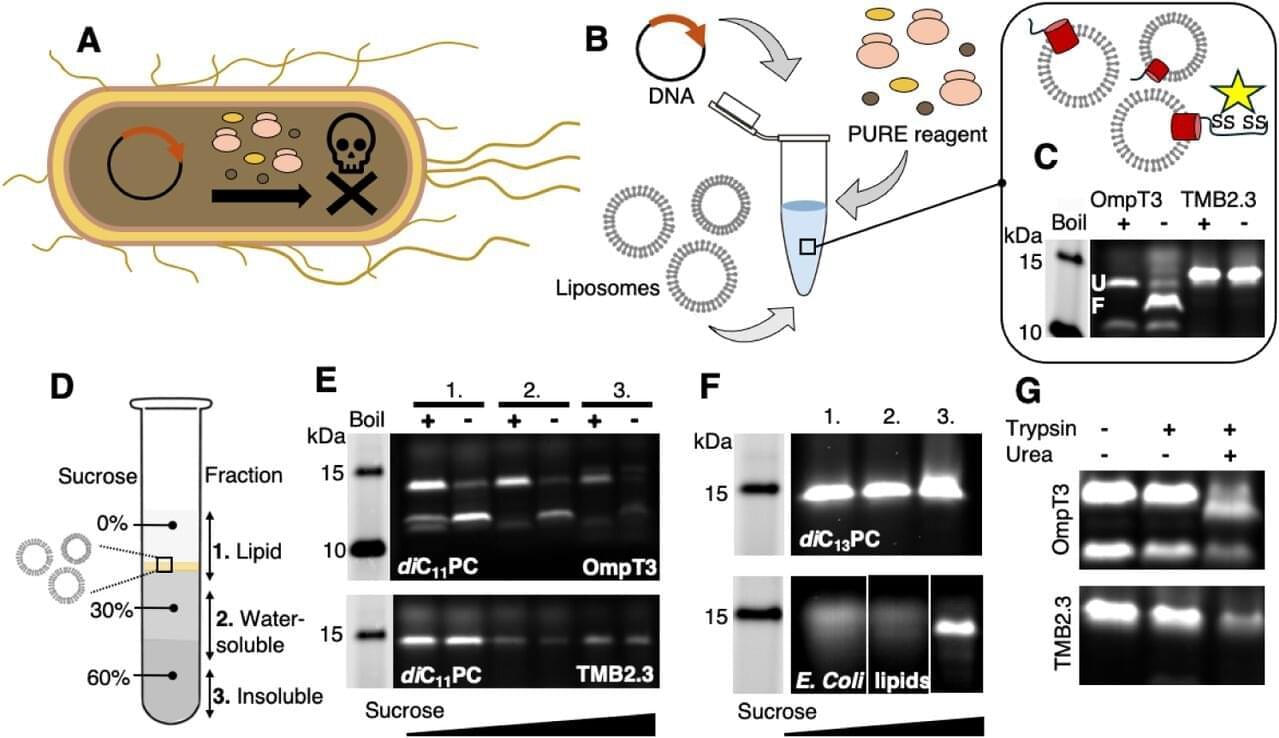
Scientists at the VIB–VUB Center for Structural Biology have uncovered a counterintuitive principle that could reshape how membrane proteins are designed from scratch: Sometimes, making a protein less stable helps it fold correctly. In their study published in the Proceedings of the National Academy of Sciences, the researchers demonstrate that introducing carefully placed “imperfections,” a strategy known as negative design, enables synthetic membrane proteins to fold and assemble efficiently in artificial membranes.
Membrane proteins are essential for life and biotechnology, acting as gateways, sensors, and drug targets. Yet designing them from scratch remains notoriously difficult. Unlike soluble proteins, they must navigate a complex folding process while inserting into lipid membranes and during this step, many designs fail.
Traditional protein design focuses on maximizing the stability of the final folded structure. But the new study shows that, for transmembrane β-barrel proteins, this approach can backfire.