Brynn Bateman unveiled the New Yokosuka project.
Get the latest international news and world events from around the world.


The Blood of Centenarians Reveals 37 Proteins Linked With Slower Aging
Science is one step closer to cracking the code of longevity thanks to a new study that identified dozens of proteins linked with slower aging in the blood of centenarians.
Scientists in Switzerland collected and compared blood samples from healthy younger individuals aged 30 to 60, hospitalized octogenarians aged 80 to 90, and centenarians aged 100 years and older, assessing how the expression of plasma proteins evolves and affects metabolism, immunity, and overall lifespan.
Of the more than 700 proteins measured, 37 formed a profile that was “closer to those of the youngest group than to those of octogenarians,” says Flavien Delhaes, cell physiologist at the University of Geneva and the study’s first author.
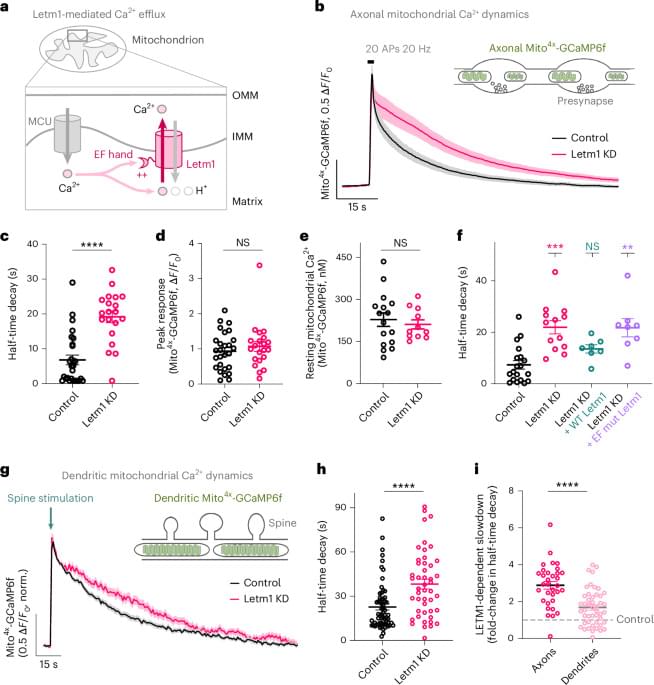
Mitochondrial Ca2+ efflux controls neuronal metabolism and long-term memory across species
Mitochondrial calcium regulates neuronal metabolism and memory.
Brain metabolism is important for long-term memories (LTMs) and various brain functions, Although it is well known that impairing neuronal metabolism limits brain performance, it is not clear if expanding the metabolic capacity of neurons boosts brain function.
In this study, the authors demonstrate that increasing mitochondrial metabolism can enhance LTM formation in flies and mice.
The authors increase mitochondrial Ca2+ by knocking down the mitochondrial Ca2+ exporter Letm1 and demonstrate over-activation of mitochondrial metabolism in neurons of central memory circuits, leading to improved LTM storage. sciencenewshighlights Science Mission https://sciencemission.com/Mitochondrial-Ca2-efflux
Boosting mitochondrial metabolism in neurons in central memory circuits by enhancing Ca2+ retention in the mitochondrial matrix is shown to improve long-term memory formation in flies and mice.

Accelerating and Streamlining Microbial Process Development
CDMO Fujifilm Biotechnologies unveiled its ShunzymeX precision purification technology, which is aimed at simplifying downstream processing for complex biologics. The technology will be presented this week at the Festival of Biologics conference in San Diego.
Fujifilm Biotechnologies, in collaboration with the University of Edinburgh, developed ShunzymeX, a proprietary technology that leverages a novel protease to enable purification of complex biologics with a simplified process, according to the company. This technology enables the addition of an affinity tag to the protein, allowing the biologic protein to be purified with an off-the-shelf affinity resin. After purification, the novel protease cleaves off the tag without leaving a scar on the native protein.
ShunzymeX addresses some of the inherent challenges in traditional microbial downstream purification due to the diversity of microbial-expressed proteins, including variability in size and sequence, leading to a lack of suitable affinity resins, says John Stewart, senior vice president of global process development, Fujifilm Biotechnologies.
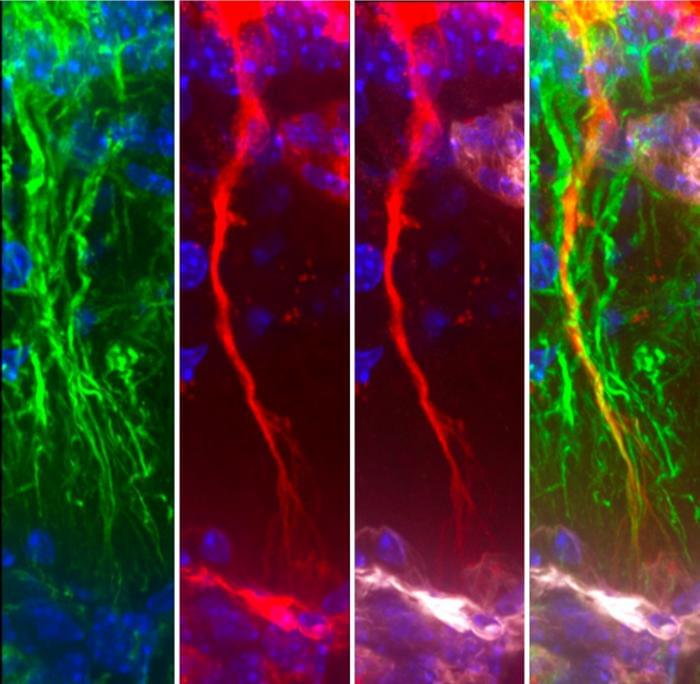
Degenerating Tanycytes Disrupt Tau Removal, Shaping Alzheimer’s Progression
“Tanycytes, whose cell bodies line the walls and floor of the third ventricle and extend long, slim processes that terminate in ‘endfeet’ that contact these fenestrated capillaries,” act as a shuttle between the CSF and the blood, the authors wrote. The new study suggests they also act as a kind of molecular “exit ramp,” moving tau out of the CSF and into the bloodstream for disposal. When these cells become fragmented, that clearance system falters. Tau, which should be ferried away, instead lingers—much like traffic backing up when a major off‑ramp closes—allowing toxic protein species to accumulate.
“Our findings reveal a previously underappreciated, disease‑relevant role for tanycytes in neurodegeneration,” said corresponding author Vincent Prévot, PhD, of INSERM. “Focusing on tanycyte health could be a way to improve tau clearance and limit disease progression.”
Using rodent and cellular models, the researchers showed that tanycytes take up tau from the CSF and release it into pituitary portal capillaries, enabling its entry into the systemic circulation, according to the authors. When the team blocked vesicular transport in tanycytes, tau clearance from CSF to blood slowed dramatically, and tau pathology intensified. As the authors wrote, “Blocking tanycytic vesicular transport blunts CSF‑to‑blood tau efflux and potentiates tau pathology.”

Can a wealthy family change the course of a deadly brain disease?
A wealthy family fighting its own disease boosted research on a little-studied brain protein, progranulin. Can it spur new dementia treatments?
Bluefield investigators, and eventually drug companies, saw something compelling about FTD-GRN, the form of the condition Alice had. In other genetic neurodegenerative disorders, such as familial Alzheimer’s and Huntington disease, mutations spark the production of toxic proteins, generating complex cascades of pathology. But the culprit mutations driving FTD-GRN block progranulin production, leaving carriers with less than half as much of the protein as noncarriers. Many dementia researchers came to describe FTD-GRN as a “low-hanging fruit” among neurodegenerative diseases, using words such as “intuitive” and “tractable” to characterize its biology. The solution seemed obvious: A treatment just needed to raise progranulin levels in the brain.
Fueled in part by that confidence, six clinical trials have been launched to test progranulin-boosting therapies in FTD-GRN. Companies also hope the anti-inflammatory properties of a progranulin-boosting agent could help in Parkinson’s disease, Alzheimer’s, amyotrophic lateral sclerosis (ALS), and FTD caused by other mutations or without a known genetic cause.
All has not gone according to plan, however. In October 2025, a landmark phase 3 clinical trial of a progranulin-boosting drug in people with FTD-GRN did not keep their disease from progressing. In February, a small trial of a gene therapy delivering a healthy copy of GRN to the brain was halted, also for lack of effect.
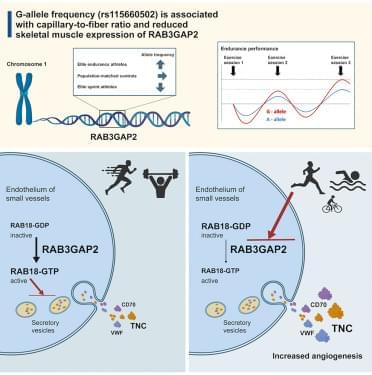
RAB3GAP2 is a regulator of skeletal muscle endothelial cell proliferation and associated with capillary-to-fiber ratio
Ström et al. identify the rs115660502 variant in RAB3GAP2 associated with increased skeletal muscle capillary-to-fiber ratio and enriched in endurance athletes. This variant reduces RAB3GAP2 expression, enhancing endothelial proliferation, tube formation, and TNC secretion, thereby promoting exercise-like angiogenesis and microvascular remodeling in skeletal muscle.