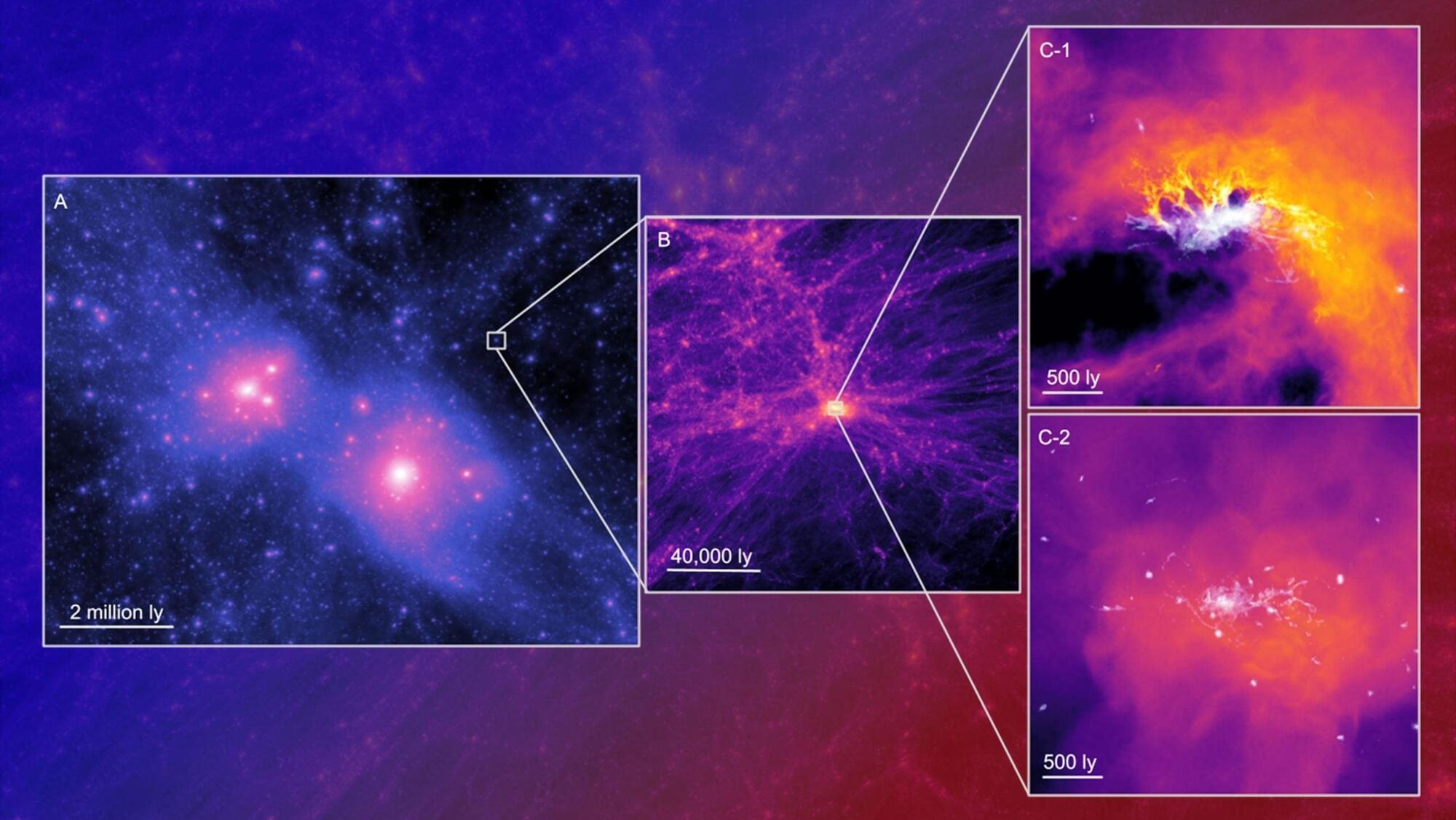
Ultra-faint dwarf galaxies are among the smallest known galaxies orbiting the Milky Way. Astronomers have long viewed them as ancient remnants from the early cosmos. Now, researchers at the Oskar Klein Centre and the LYRA collaboration have used a powerful new set of simulations to show that these dim galaxies may reveal how conditions in the young Universe shaped which galaxies were able to grow and which never formed stars at all.
The study, published in Monthly Notices of the Royal Astronomical Society (MNRAS), was led by Azadeh Fattahi, Associate Professor at the Oskar Klein Centre (OKC), along with collaborators from Durham University and the University of Hawaii.
She explains the scale of the project: “In this work we presented a brand-new suite of cosmological simulations focused on the faintest galaxies in the Universe, with an unprecedented resolution. These are by far the largest sample of such galaxies ever simulated at these resolutions.”