The immune system’s killer T cells do a commendable job of detecting and destroying cancer cells. But the harsh environment at the heart of tumors often saps them of their vitality, pushing them into a state of permanent lassitude called “terminal exhaustion.” The phenomenon accounts for why so many tumors resist routine immune clearance and even cancer immunotherapies devised to stimulate their lethal capabilities.
Terminal exhaustion is characterized by an accumulation of dysfunctional mitochondria—the bean-shaped energy generators in cells—and extensive genetic reprogramming that stalls proliferation and hobbles the cell-killing weaponry of T cells. Yet how mitochondrial dysfunction is linked to genetic reprogramming in the cells was unclear. No longer. Researchers in the journal Nature show that how the accumulation of useless mitochondria is linked to T cell exhaustion through a complex series of subcellular processes.
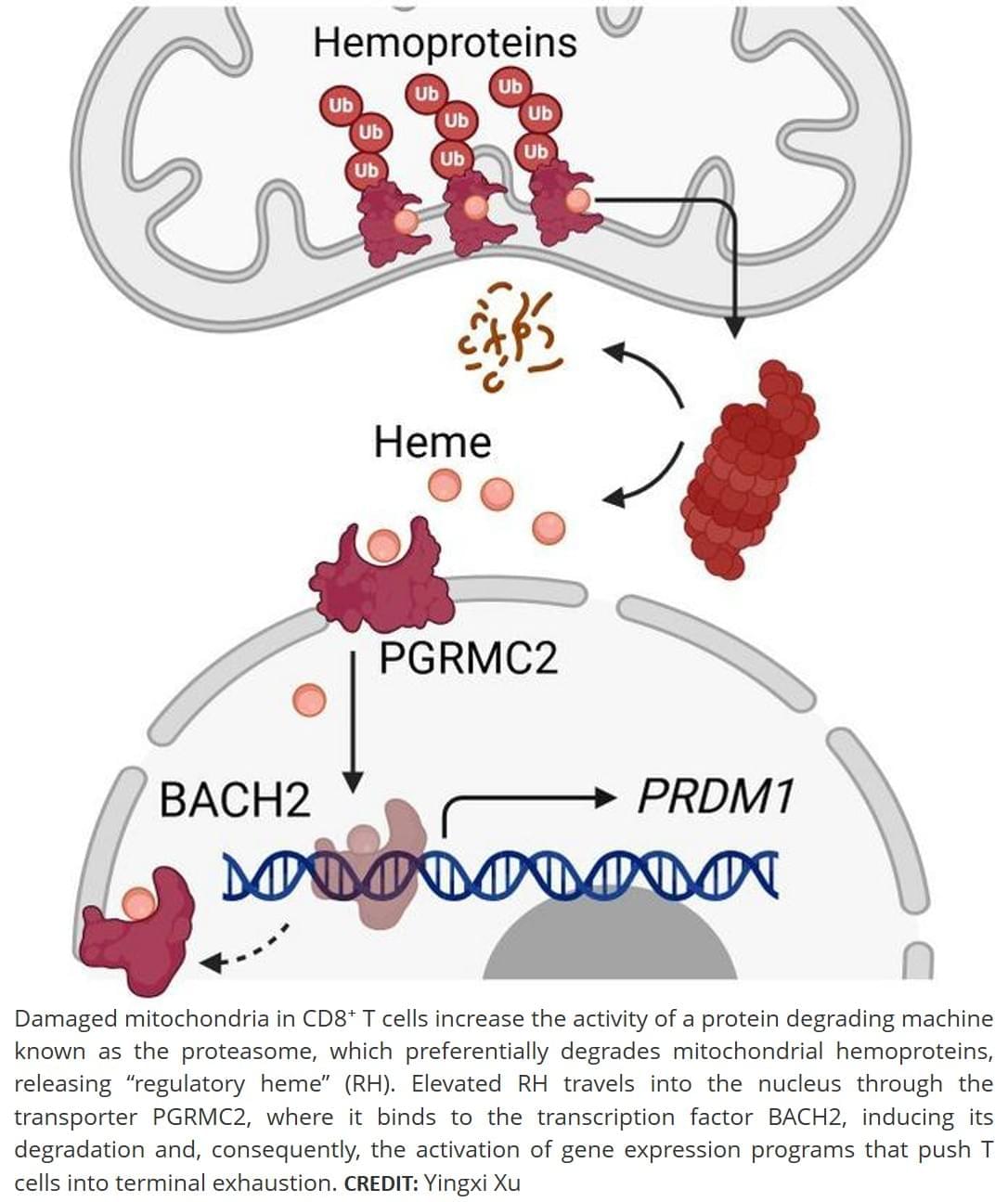
The researchers report in their paper that the glut of dysfunctional mitochondria enhances the activity of a cellular protein digesting machine, known as the proteasome, in T cells. The activated proteasome, they show, preferentially degrades mitochondrial heme-containing proteins.
As might be expected, this bias leads to quite the buildup of heme in the cells, resulting in the generation of a functionally distinct form of the molecule referred to as “regulatory heme,” which zips into the nucleus through a transporter named PGRMC2. There it binds to a transcription factor, a protein that regulates gene expression, causing its degradation. This kicks off a series of events that culminates in the activation of genetic programs known to induce terminal exhaustion.
The researchers show that genetic disruption of PGRMC2 abrogates this effect, keeping anti-tumor T cells in a functionally vibrant state, suggesting it is a potential drug target for the enhancement of T cell-activating cancer immunotherapies.
The researchers also examined how the pharmacological inhibition of the proteasome with an existing leukemia therapy, bortezomib, might affect CAR-T cells. Like bortezomib, CAR-T therapy is currently used to treat B cell acute lymphoblastic leukemia (B-ALL).
“We found that the transient and low-dose addition of bortezomib to CAR-T cell cultures during manufacturing reduces exhaustion-associated programs in the cells and induces durable reprogramming of their gene expression patterns to maintain them in a proliferative and functionally vibrant state,” said the author. ScienceMission sciencenewshighlights.