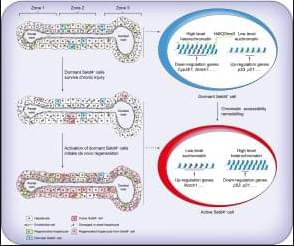
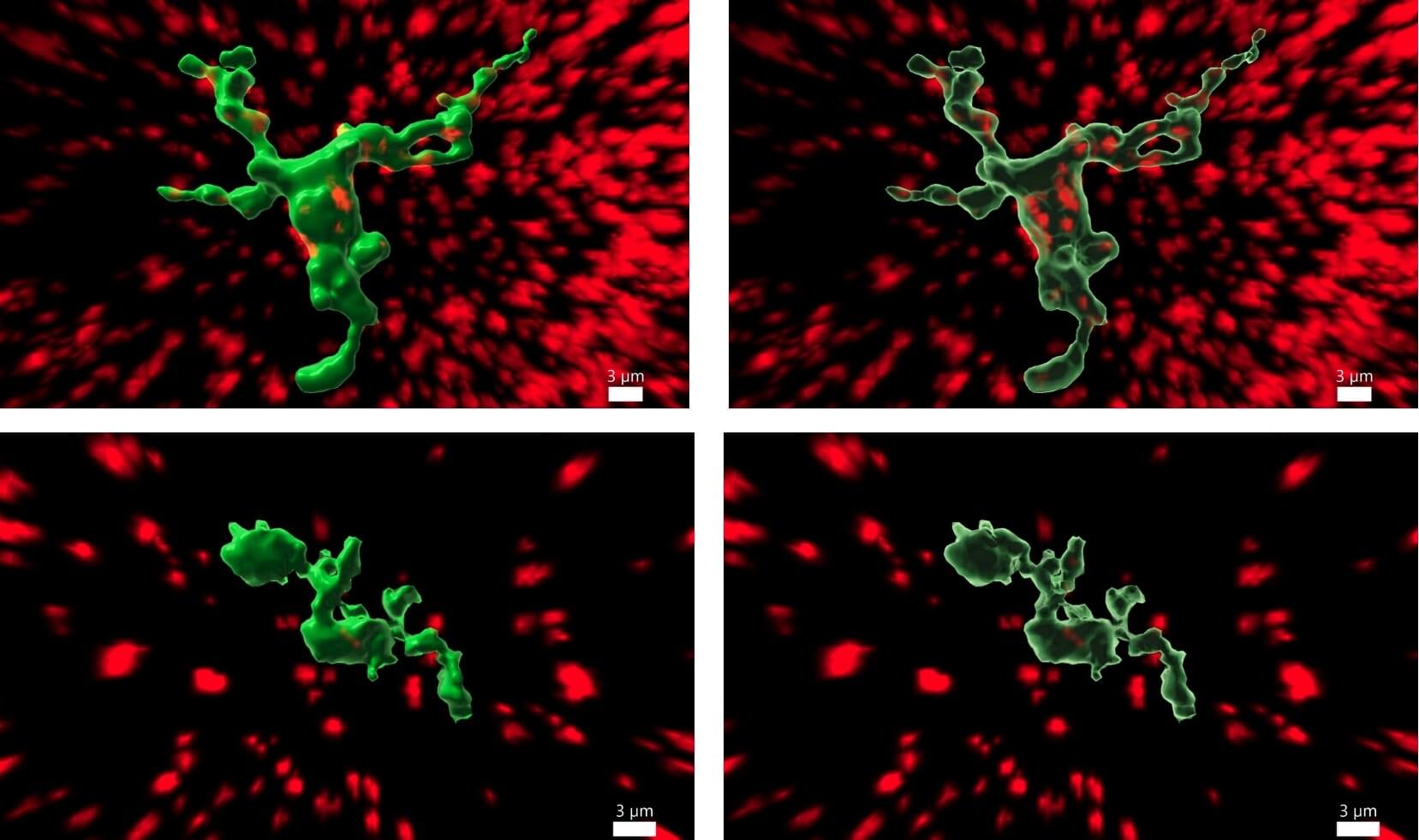
Liver regeneration in chronic disease is incompletely understood. In this issue, Jia and coworkers identify a distinct population of hepatocytes expressing Setd4 as key contributors to liver regeneration after chronic injury. Using lineage-tracing and ablation strategies in mouse models of chronic liver damage due to thioacetamide or 3,5-diethoxycarbonyl-1,4-dihydrocollidine diet, the authors show that Setd4+ cells are a long-lasting, dormant, injury-resistant hepatocyte population that survives chronic insults and get activated to support de novo regeneration. Quiescent Setd4+ cells show suppressed metabolic activity maintained by a chromatin state that promotes cellular dormancy and survival (H4K20me3-enriched heterochromatin). After chronic injury, chromatin accessibility remodelling occurs, leading to activation of the cells and initiating regeneration. Selective ablation of Setd4+ cells markedly impaired regenerative recovery, leading to increased fibrosis, higher transaminases, and shorter survival. Interestingly, Setd4+ cells appeared to be essential to initiate de novo regeneration under chronic but not acute conditions. Given the presented data, this newly identified dormant Setd4+ hepatocyte population might hold therapeutic potential to restore regeneration in chronic liver disease.
Full text here: https://www.journal-of-hepatology.eu/article/S0168-8278(…4/fulltext.
EASL — the home of hepatology.
Chronic liver injury and its progression to disease often extend beyond exposure to toxic metabolites or xenobiotics. Recovery from chronic injury, when achieved, depends on de novo regeneration, the underlying mechanisms of which remain poorly understood. Herein, we investigate a specific cell population proposed to be fundamental for de novo regeneration and recovery following chronic injury, aiming to elucidate its regulatory mechanisms.