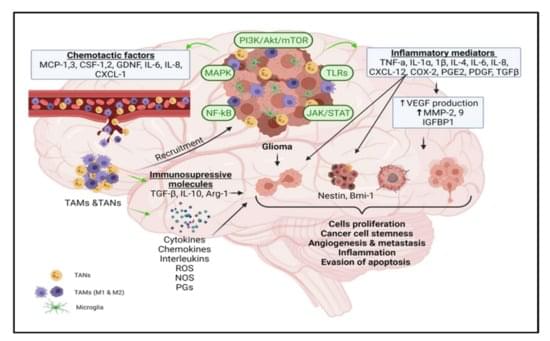
Simple SummaryThe tumor microenvironment is a complex network comprised of neoplastic and a variety of immune cells, proteins, and inflammatory mediators.


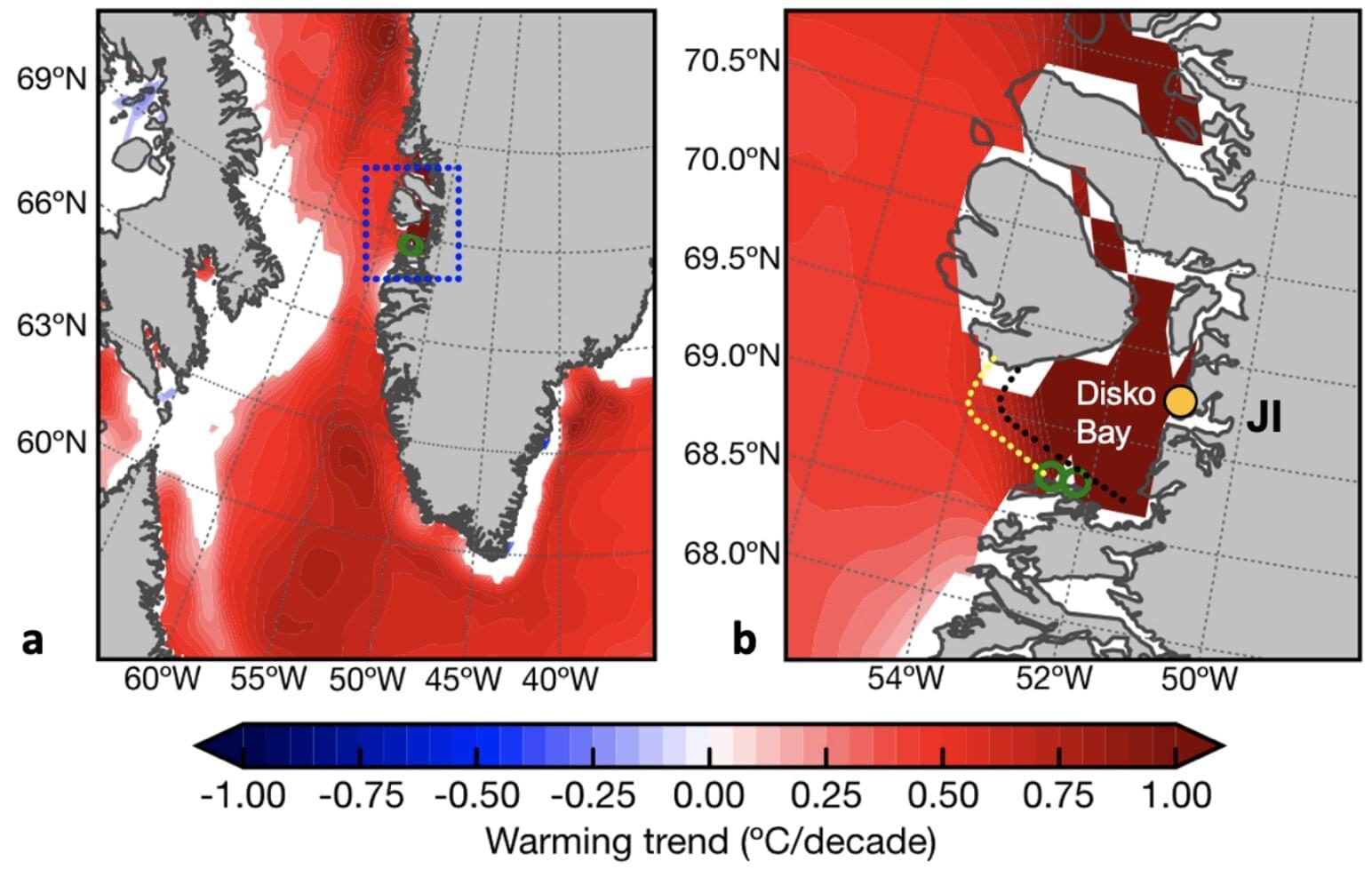
Greenland’s largest glacier, Jakobshavn Glacier, may be edging closer to a critical threshold as meltwater runoff from the Greenland Ice Sheet accelerates in ways not seen in over a century, according to new research published in Climate of the Past. The study reconstructs more than 100 years of freshwater discharge flowing from the ice sheet into Disko Bay in western Greenland, revealing a striking and sustained change that began in the early 2000s.
Researchers from Kiel University, Germany, and colleagues found that runoff did not increase gradually, but instead shifted into sharp acceleration. By 2007, the volume of freshwater entering the ocean had permanently exceeded the range of natural variability seen throughout the 20th century. Simply put, the system appears to have moved into a new state, one characterized by consistently higher meltwater output. This pattern suggests the ice sheet may be approaching what scientists call a “tipping point”—a threshold beyond which changes become self-reinforcing and potentially difficult to reverse.
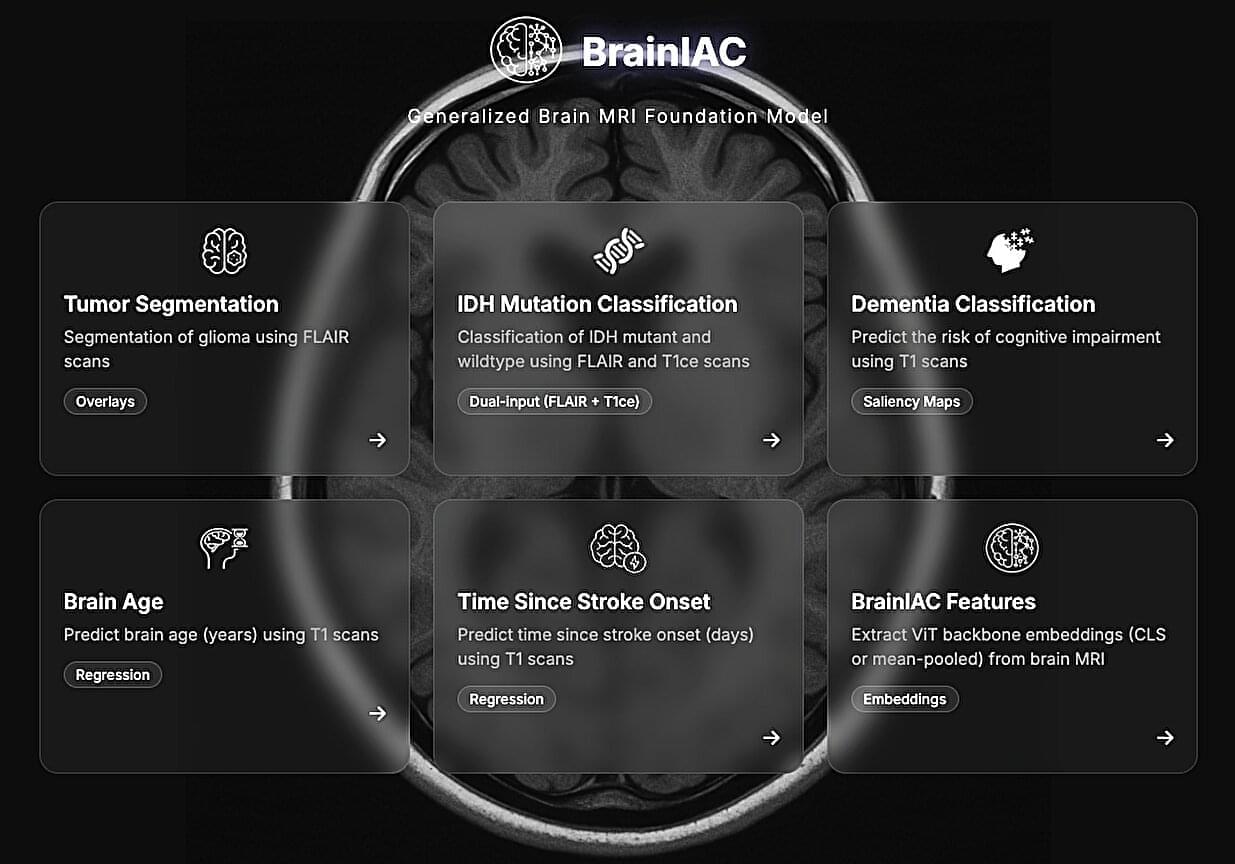
Artificial intelligence (AI) systems are computational models that can learn to identify patterns in data, make accurate predictions or generate content (e.g., texts, images, videos or sound recordings). These models can reliably complete various tasks and are now also used to carry out research rooted in different fields.
Over the past few decades, some AI models have proved promising for the early diagnosis and study of specific diseases or neuropsychiatric conditions. For instance, by analyzing large amounts of brain scans collected using a noninvasive technique known as magnetic resonance imaging (MRI), AI could uncover patterns associated with tumors, strokes and neurodegenerative diseases, which could help to diagnose these conditions.
Researchers at Mass General Brigham, Harvard Medical School and other institutes recently developed Brain Imaging Adaptive Core (BrainIAC), a large AI system pre-trained on a vast pool of MRI data that could be adapted to tackle different tasks. This foundation model, presented in a paper published in Nature Neuroscience, was found to outperform many models that were trained to complete specific medical or neuroscience-related tasks.

The evolution of eukaryotes was accompanied by an increased need for intracellular communication and cellular specialization. Thus, a more complex collection of secreted and membrane proteins had to be synthesized, modified, and folded. The endoplasmic reticulum (ER) thereby became equipped with devoted enzymes and associated factors that both catalyze the production of secreted proteins and remove damaged proteins. A means to modify ER function to accommodate and destroy misfolded proteins also evolved. Not surprisingly, a growing number of human diseases are linked to various facets of ER function. Each of these topics will be discussed in this article, with an emphasis on recent reports in the literature that employed diverse models.
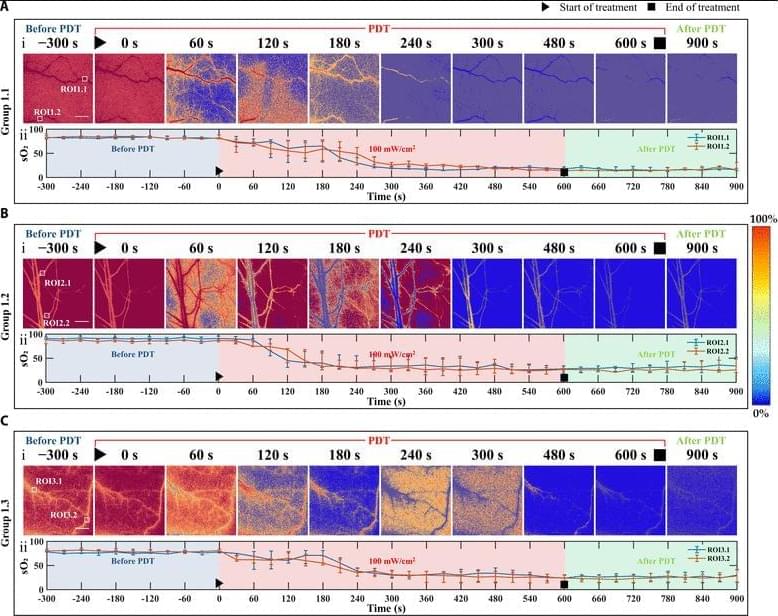
Objective: To enhance vascular-targeted photodynamic therapy (V-PDT) efficacy by integrating real-time dosimetric monitoring and adaptive irradiance modulation based on dynamic physiological feedback. Impact Statement: This study presents a closed-loop, dual-modality optical imaging-guided V-PDT platform that enables individualized, oxygen-informed irradiance control, improving therapeutic precision and efficiency. Introduction: While V-PDT is a promising, minimally invasive treatment for tumors and vascular abnormalities, its efficacy is often hindered by rapid oxygen depletion under high irradiance, leading to treatment-limiting hypoxia. Accurate, real-time assessment of both photosensitizer concentration and blood oxygenation is essential to guide optimized therapeutic strategies, yet such capability has remained elusive in clinical settings.
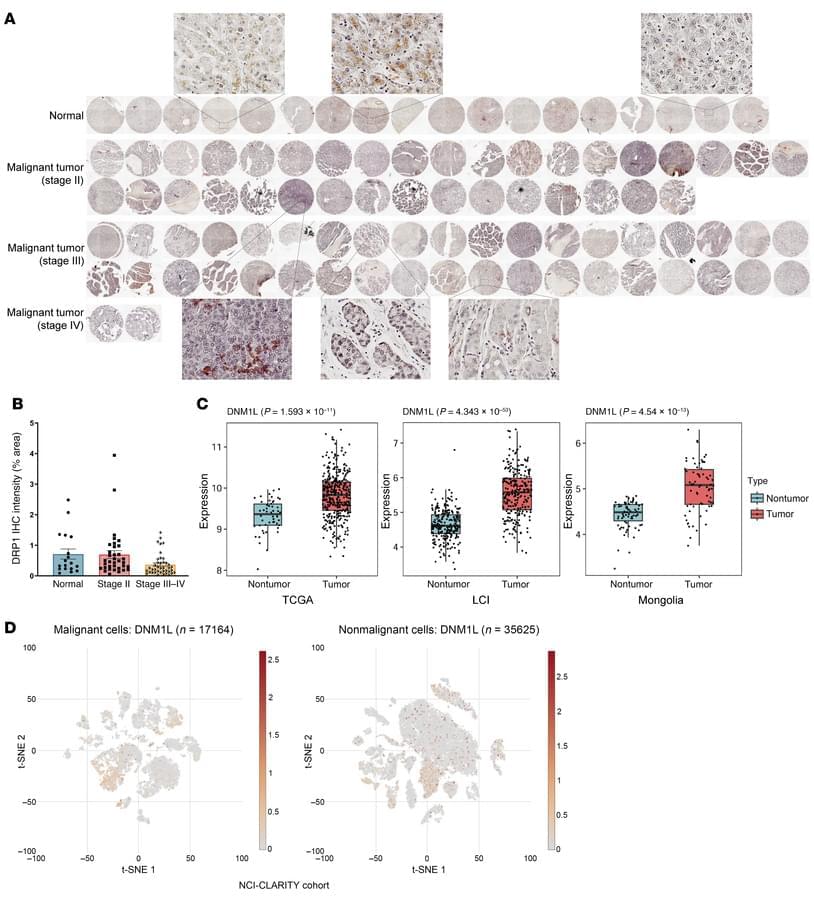
Here, Wen-Xing Ding find alterations in mitochondrial dynamics and the accumulation of large mitochondria contribute to liver tumor development in mice: https://doi.org/10.1172/JCI194441 # MASH
The EM image shows liver cells with megamitochondria (arrows) from mice lacking liver-specific dynamin-related protein 1 (Dnm1).
1Department of Pharmacology, Toxicology and Therapeutics, University of Kansas Medical Center, Kansas City, Kansas, USA.
2Department of Pharmaceutical Sciences, Eugene Applebaum College of Pharmacy and Health Sciences and.
3Department of Pharmacology, School of Medicine, Wayne State University, Detroit, Michigan, USA.

Scientists at the University of California, Irvine’s School of Pharmacy & Pharmaceutical Sciences have discovered how muscle stem cells “flip a switch” to rebuild damaged muscle—a finding that could help address muscle loss linked to aging, injury and widely used weight-loss medications.
The study, published this week in Nature Metabolism, shows that muscle recovery is not just about protein or exercise. It depends on timing and how muscle cells use fuel.
Researchers learned that immediately after stress, muscle stem cells temporarily slow down energy production. Instead of burning glucose for energy, they reroute it into protective repair processes to produce antioxidants that reduce inflammation. Once repairs are complete, energy production ramps back up and new muscle fibers form and strengthen.