Nasal tissue harbours T cells that ‘remember’ a pathogen long after infection is past.


Space has become critical infrastructure for climate monitoring, disaster risk reduction, connectivity, navigation, education, and long-term planetary resilience. Even more important, space is an open horizon for new industrial development and settlement, starting with Earth orbit, the geo-lunar system, and the near-Earth asteroids. The Space 18th SDG initiative proposes a non-regulatory, enabling framework that strengthens the existing 17 SDGs by recognizing outer space as both an enabler of sustainable development and an environment requiring stewardship.
THE PANEL:
Prof. Sergio Marchisio, Space Law Expert, La Sapienza University, Rome, Italy.
Ms. Fikiswa Majola, Deputy Director Space Systems, Department of Science and Technology (DST) South Africa.
Prof. Guoyu Wang — Space Law Center, China National Space Administration.
Dr. Claire Nelson, The Future Forum, Giamaica.
Adriano V. Autino, SRI CEO & Founder.
Maria Antonietta Perino, Thales Alenia Space, Italy.
Stefano Antonetti, D-ORBIT SpA, Strategy Director, Italy.
Antonio Stark, iSpace, Japan.
MODERATES:
Dr. Gülin Dede, SRI Director of Relations, Chair of the Space 18th SDG Coalition.


Researchers at Oregon Health & Science University have uncovered a key reason why immunotherapy has largely failed in pancreatic cancer—and identified a promising strategy to overcome that resistance. The study, published in the journal Immunity, shows that pancreatic tumors actively reshape their immune environment by co-opting regulatory immune cells that normally shut down tumor-killing cells. By reprogramming those cells, the research reveals a potential pathway to make immunotherapy effective against one of the deadliest and most treatment-resistant cancers.
“Pancreatic cancer is incredibly resistant to most therapies,” said the study’s senior author, Katelyn Byrne, Ph.D., assistant professor of cell, developmental and cancer biology in the OHSU School of Medicine and member of the OHSU Brenden-Colson Center for Pancreatic Care. “Even when we know the immune system is capable of long-lasting protection, it’s been very difficult to get that response to work in this disease.”
Immune checkpoint inhibitors and other immunotherapies have transformed care for cancers such as melanoma and lung cancer, but they have shown little benefit for pancreatic cancer. One major reason, Byrne said, is the presence of large numbers of regulatory T cells, or Tregs, inside pancreatic tumors.
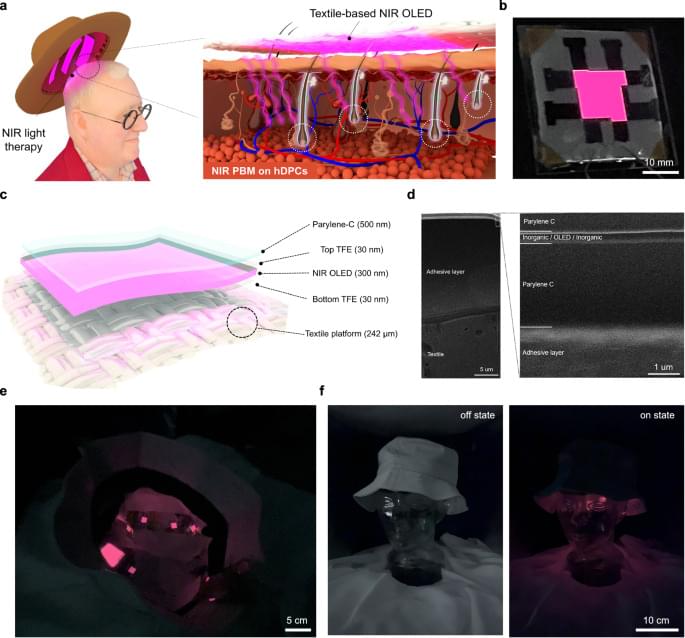
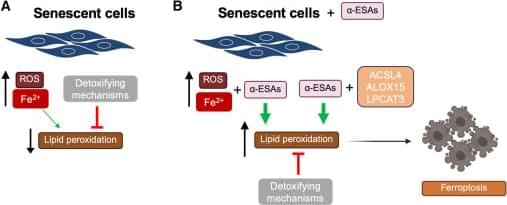
In a recent Cell Press Blue paper, Zhang et al. identify two polyunsaturated lipids that selectively eliminate senescent cells by inducing ferroptosis, uncovering this iron-dependent cell death pathway as a vulnerability for senescent cells. Their findings position ferroptosis induction as a promising strategy for targeting senescence and aging-associated diseases.
Researchers at Oregon State University have pioneered a transformative approach for simultaneously targeting lung cancer and the debilitating muscle-wasting syndrome known as cachexia—a condition that plagues many lung cancer patients. Their groundbreaking work employs lipid nanoparticles (LNPs) as a delivery vehicle for messenger RNA (mRNA) therapeutics, addressing critical challenges in precision drug delivery for aggressive tumors deep within the lung tissue.
Lipid nanoparticles, microscopic carriers composed of fatty compounds like lipids, have revolutionized drug delivery with their ability to ferry genetic material directly into cells. In this study, the OSU team engineered LNPs comprised of DC-cholesterol and a specialized ionizable lipid, 113-O12B, which exhibited a remarkable ability to bind a blood serum protein called vitronectin. This binding triggers the formation of a protein corona on the nanoparticles, a dynamic interface that actively guides the LNPs to lung tissue, and more importantly, lung tumor microenvironments.
Vitronectin’s recruitment is no coincidence. It interacts with integrin receptors—cellular docking proteins highly expressed on lung cancer cells. These integrins act as biological gateways, facilitating enhanced uptake of the therapeutic nanoparticles by tumor cells while sparing healthy tissue. This receptor-mediated targeting marks a significant advance over conventional LNPs, which commonly accumulate in the liver, limiting their therapeutic index against lung malignancies.
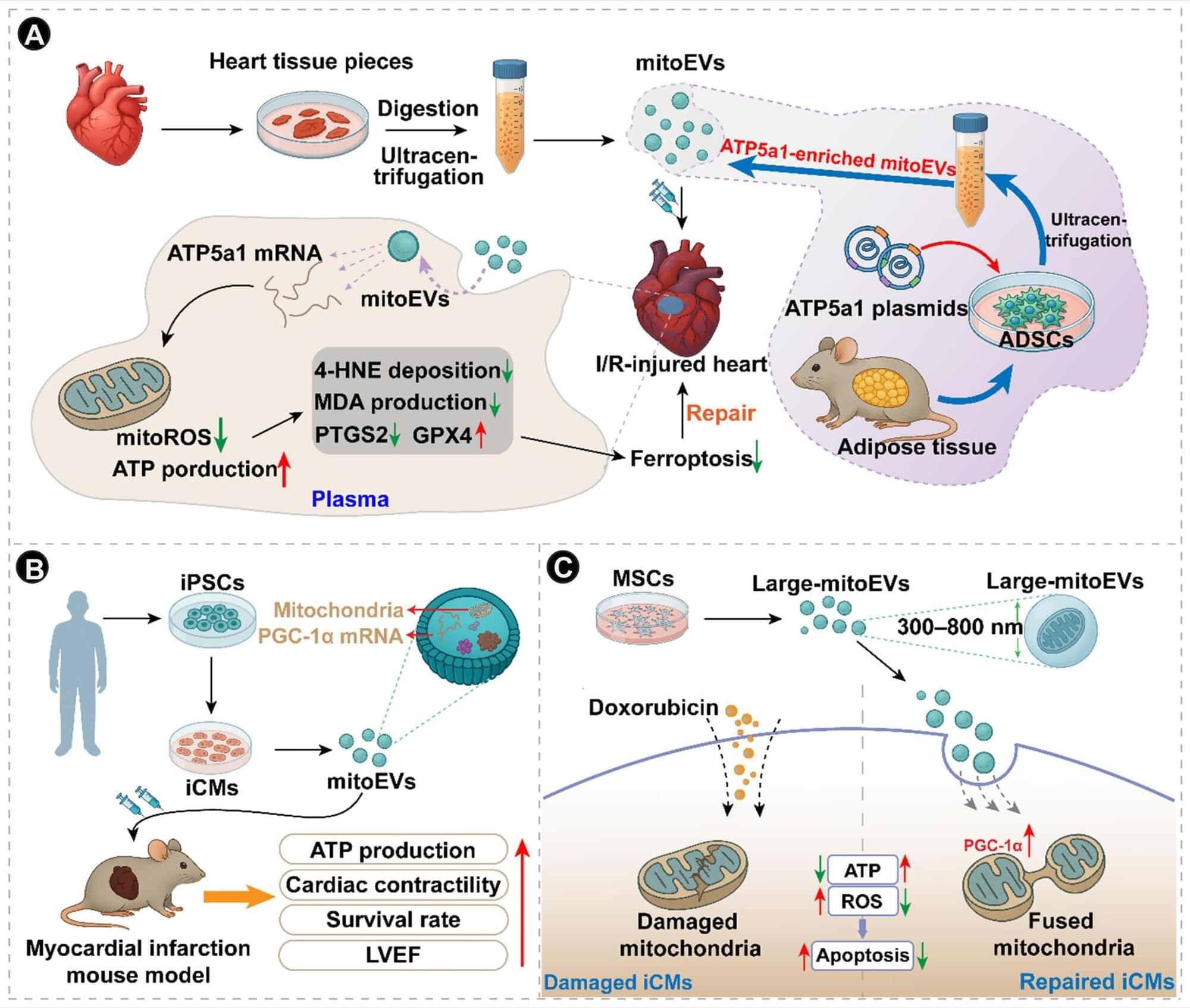
@CircRes Compendium on Migration of Mitochondria Beyond Cell Boundary.
Authored by Drs. Rapushi & colleagues.
Mitochondria-derived vesicles (MDVs) and mitochondrial extracellular vesicles (mitoEVs) represent 2 related extensions of mitochondrial dynamics that link organelle maintenance to communication within and between cells. MDVs are small vesicles that bud directly from mitochondria, selectively packaging components of the outer membrane, inner membrane, or matrix. They serve as a localized quality control mechanism that removes oxidized or damaged material without engaging the entire mitophagic machinery. After budding, MDVs typically enter the endolysosomal pathway, where they can fuse with late endosomes or lysosomes for cargo degradation. A subset of MDVs also targets other organelles, particularly peroxisomes, contributing to organelle crosstalk, lipid metabolism, and redox balance.
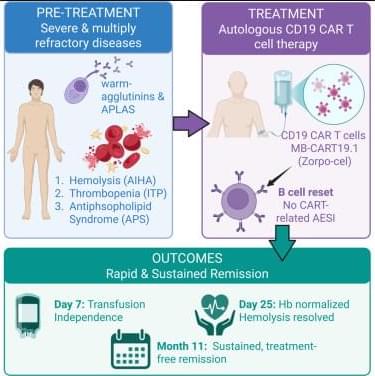
Cutting edge CAR-T cell therapy drives remission of three life-threatening autoimmune diseases in patient.
Med by Cell Press.
This single-patient case report expands the indications in which CD19 CAR-T cell therapy demonstrates unprecedented clinical efficacy, achieving sustained, treatment-free remission. Following B cell abrogation, AIHA is stopped, antiphospholipid antibodies are abrogated, and an underlying ITP is stabilized without any CAR-typical side effects.