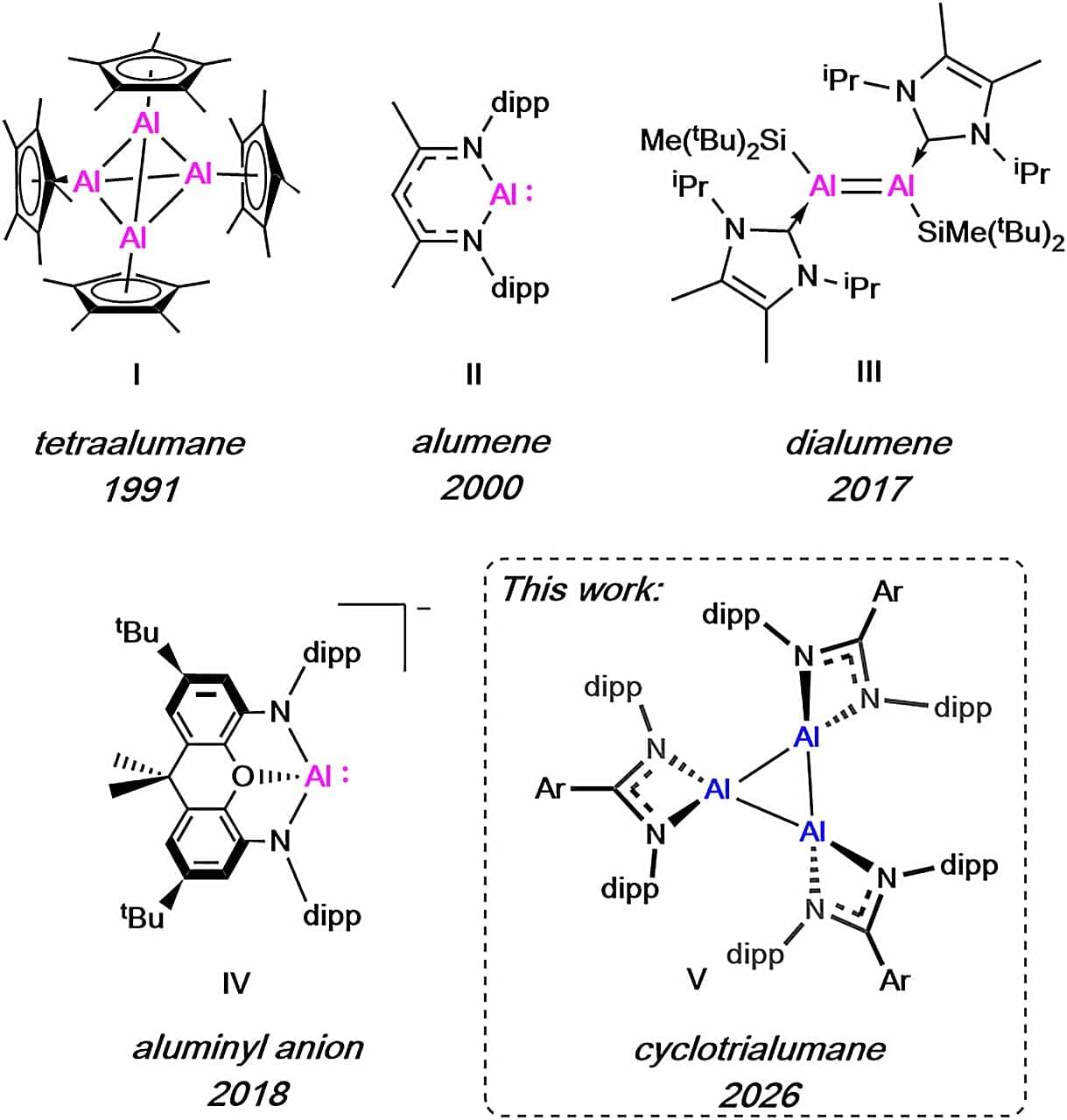
A research team at King’s College London has isolated a new form of aluminum—a highly abundant metal, that could provide a far cheaper and more sustainable alternative to commonly used rare earth metals. Dr. Clare Bakewell, Senior Lecturer in the Department of Chemistry, and her lab developed highly reactive aluminum molecules able to break apart tough chemical bonds. Published in Nature Communications, their work has also unlocked molecular structures that have never been observed before, which creates the potential for new kinds of reactive behavior.
The team reported the first example of a cyclotrialumane, a compound comprising three aluminum atoms arranged in a trimeric—triangular—structure. The trimeric molecule carries unprecedented reactivity as the structure is retained when dissolved into different solutions, making it robust enough for use in a range of chemical reactions. These include splitting dihydrogen and the stepwise insertion and chain growth of the 2-carbon hydrocarbon, ethene.
Metals are vital for making a whole range of commodity and fine chemicals produced in industry. However, many processes, especially catalytic ones, use expensive precious materials like platinum, which are environmentally damaging to extract.