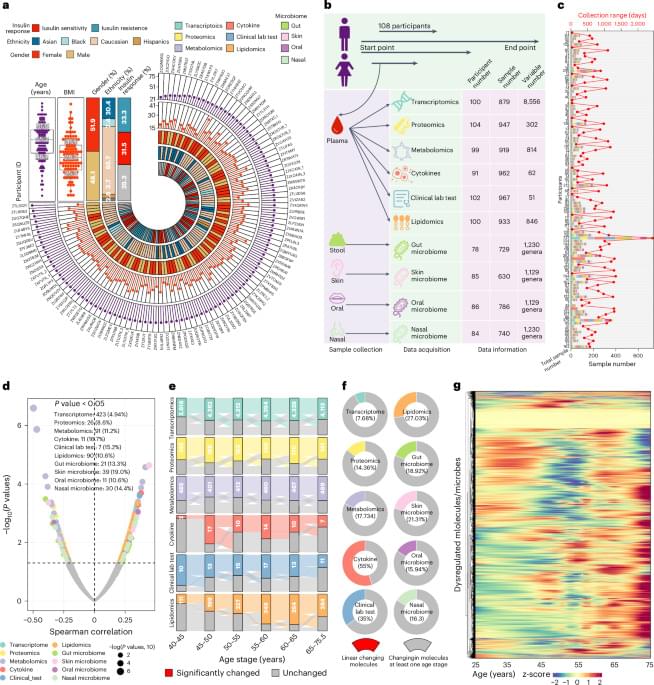
Researchers found that removing certain inflammation-causing cells from mice every month, starting in their middle age, not only extended their lifespans but also improved their heart health and overall physical function throughout their lives. This method shows promise for extending the period of good health as mice age, potentially pointing to new aging treatments for humans.
Everyone wants to live to a ripe old age, but no one wants to be decrepit. Now, University of Connecticut researchers have demonstrated a treatment that could lengthen life—and vigor—up to the very end.
Even as human lifespans have lengthened over the past century, most people in old age suffer a serious health decline in the last decade of life. Chronic illnesses such as cancer, diabetes, or cardiovascular disease may begin, followed by frailty. Many interventions can prolong life, but not necessarily good health. And nobody wants to spend the last years of an extra-long life in decrepitude.