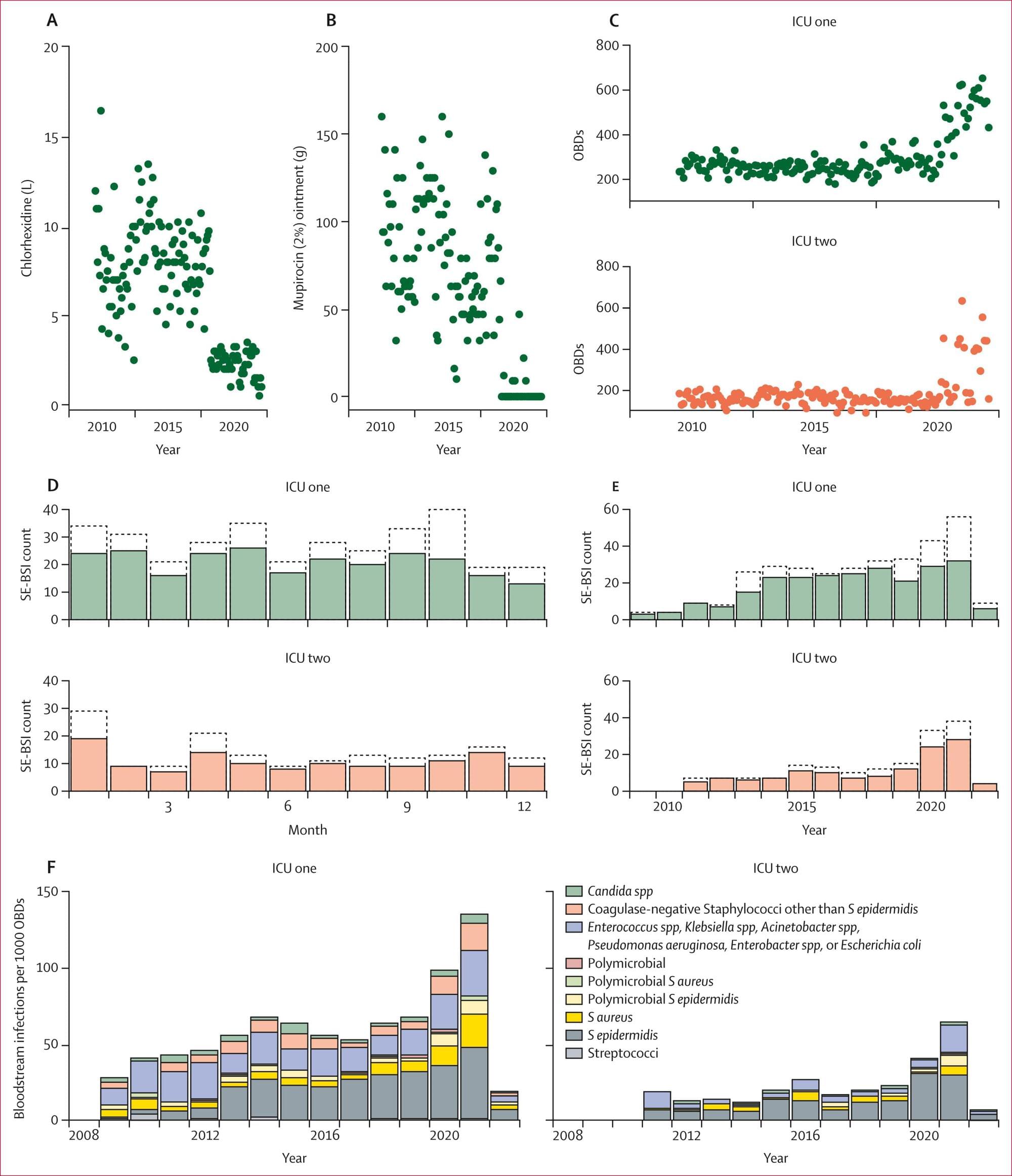
An international study has, for the first time, revealed a strong and direct link between the rise of antibiotic-resistant bacteria and universal disinfection procedures applied to patients in intensive care units. Published in The Lancet Microbe, the study calls for a reassessment of health care guidelines on the widespread use of disinfectants.
“Our research highlights the unintended consequences of universal decolonization in a global context where antibiotic resistance is an increasing threat,” says Marco Oggioni, professor at the Department of Pharmacy and Biotechnology at the University of Bologna and one of the study’s authors. “Coordinated efforts to prevent antibiotic-resistant infections are crucial, but they must not prevent us from critically re-evaluating the tools we use to achieve these goals.”
Universal decolonization is a preventive procedure applied to patients upon admission to intensive care. The entire body is disinfected with chlorhexidine—a commonly used antiseptic also employed to sanitize medical devices and hospital surfaces—and an additional nasal treatment is administered with another disinfectant, mupirocin.