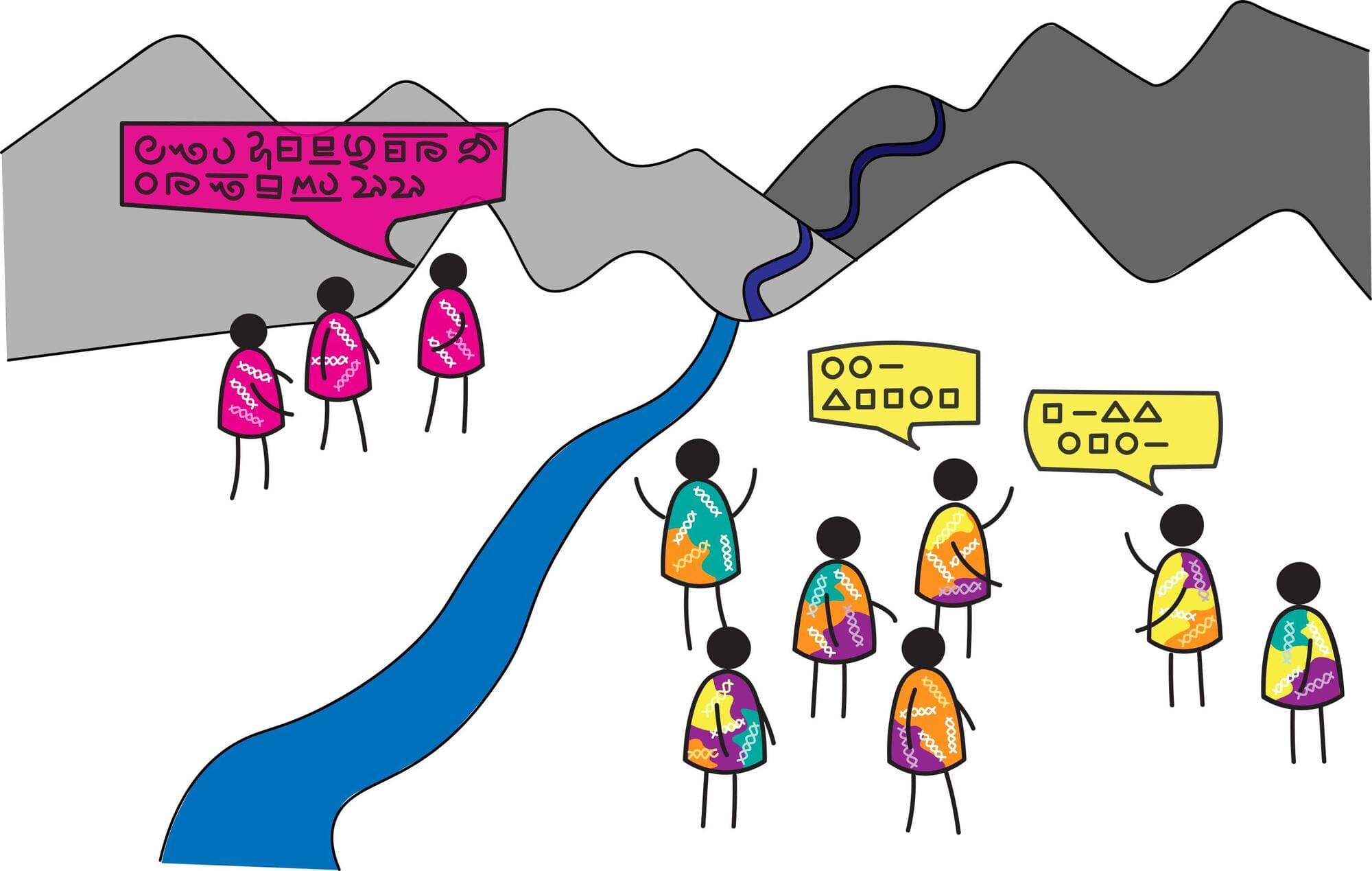
Languages and human DNA both capture aspects of human diversity. But how are they related? A new international study led by the University of Zurich finds a clear but counterintuitive pattern: regions with high genetic diversity tend to have more similar languages, while isolated populations with low genetic diversity show greater linguistic diversity. The research is published in the journal Proceedings of the National Academy of Sciences.
At first glance, the findings seem surprising. One might expect regions with greater genetic diversity, often shaped by migration and population mixing, to also show greater diversity in language. But the study reveals the opposite.
“We were struck by how robust this inverse relationship is across the globe,” says Anna Graff, lead author of the study and linguist at the University of Zurich. “Places where people have mixed more tend to be genetically diverse, but their languages are structurally more similar. In contrast, places with long-term isolation show less genetic diversity, yet much greater diversity in how languages are structured. Crucially, this relationship holds after adjusting for a wide range of confounding factors, including deep population history such as the timing of continental settlement.”