In patients with atrial fibrillation and recent stroke, adding catheter ablation to oral anticoagulation did not significantly reduce recurrent stroke, systemic embolism, death, or hospitalization for heart failure compared to standard therapy.
This randomized clinical trial found no significant difference between the standard-therapy group and the ablation group in the primary composite end point, which included recurrent ischemic stroke, systemic embolism, all-cause death, and hospitalization for heart failure.
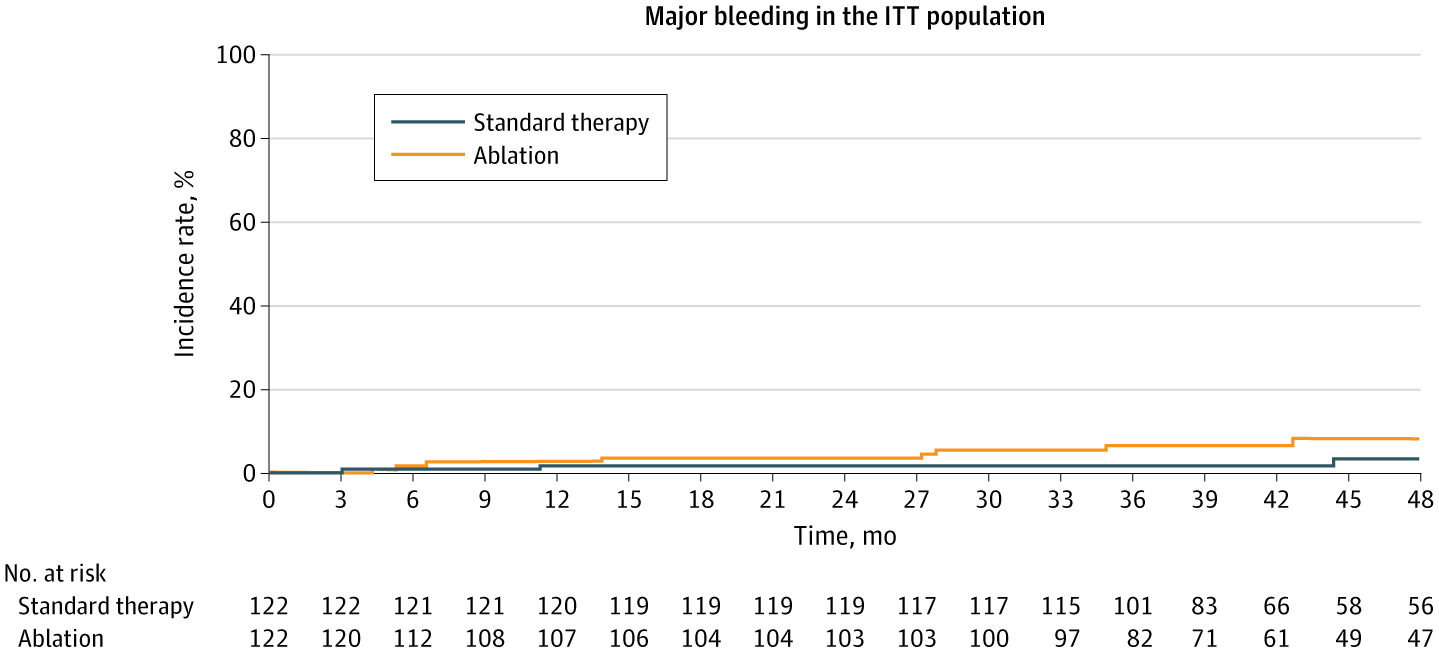
The incidence rate of recurrent ischemic stroke in the standard-therapy group was 3.1 per 100 person-years, which was lower than previously reported values.22-24 Conversely, the incidence of recurrent ischemic stroke in the ablation group was 2.5 per 100 person-years, which is consistent with a recent report.25 Although the difference in the incidence rate of recurrent ischemic stroke between the 2 groups was not statistically significant, it remains possible that catheter ablation has a beneficial effect on reducing recurrent ischemic stroke. The lower-than-expected rate of recurrent ischemic stroke in the standard-therapy group likely reflected the appropriate continuation of direct oral anticoagulants in all patients, the exclusion of patients with severe stroke, recent advances in atrial fibrillation treatment, and the comanagement of atrial fibrillation care by neurologists and cardiologists.
All-cause death occurred in 5 patients (1.0 per 100 person-years) in the standard-therapy group and 12 patients (2.8 per 100 person-years) in the ablation group. The mortality rate in both groups was lower than expected, which is likely explained by the exclusion of patients with severe stroke, active cancer, or markedly impaired cardiac function. In most patients who died, more than 1.5 years had elapsed between the catheter ablation procedure and death, suggesting that most deaths were unlikely to be directly related to the catheter ablation procedure itself. In this study, patients in the ablation group more frequently had a history of heart disease, and the incidence rates for major bleeding and cardiovascular death were higher than those for patients in the standard-therapy group, which may be associated with the increased mortality observed in this group.26 However, the underlying reasons for this observation remain unclear.