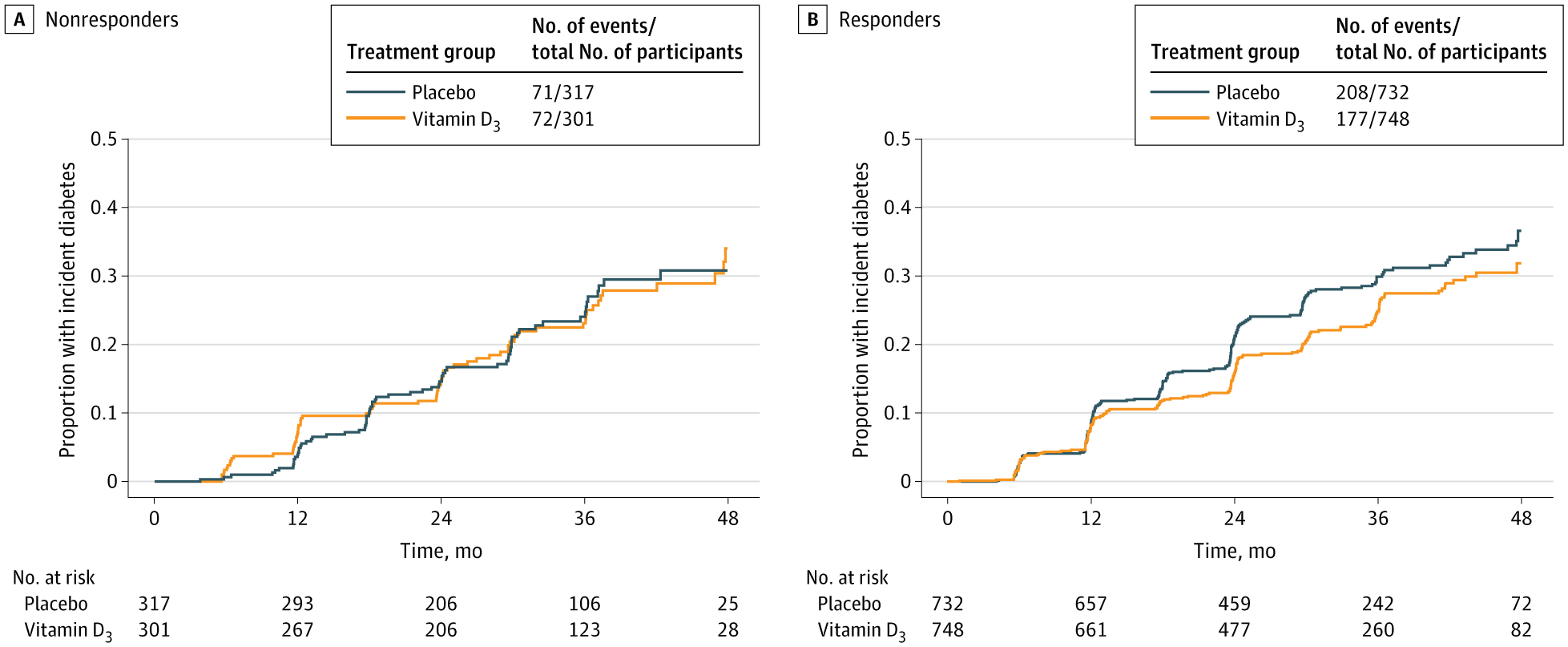
Among adults with Prediabetes, vitamin D3 supplementation was associated with lower diabetes risk only in those with specific ApaI vitamin D receptor genotypes.
This genetic association analysis of the D2d study suggests that genetic variation in the VDR, specifically the ApaI polymorphism, is associated with diabetes risk at higher intratrial 25(OH)D levels and is associated with response to 4,000 IU/d of vitamin D3 supplementation among adults with prediabetes. Participants carrying the ApaI AA genotype did not experience a reduction in diabetes risk, either when achieving higher intratrial 25(OH)D concentrations or while being treated with 4,000 IU/d of vitamin D3. In contrast, those carrying the ApaI CC and AC genotypes, representing 71% of the D2d study population, had progressively lower risk of type 2 diabetes at intratrial 25(OH)D levels of 40 ng/mL or higher. Participants with these genotypes randomized to vitamin D3 had a 19% reduction in the risk of progression to diabetes compared with placebo, whereas those with ApaI AA alleles did not respond to treatment with vitamin D3. The BsmI polymorphism also appeared to play a role in the association between the achieved intratrial 25(OH)D level and diabetes risk, as expected given the high linkage disequilibrium of ApaI and BsmI (D’ = 1.0 and r2 = 1.0) among people of European ancestry.15 Because there was a near complete overlap between participants carrying the nonresponsive Bsml TT genotype and those carrying the nonresponsive ApaI AA genotype, knowing the ApaI genotype alone was sufficient to identify individuals who were likely—or unlikely—to respond to supplementation with 4,000 IU/d of vitamin D3. These exploratory genetic association findings support our hypothesis that a common VDR variant modulates the link between high intratrial 25(OH)D levels and diabetes risk, and the association between relatively high-dose vitamin D3 supplementation and diabetes risk among adults with prediabetes. The distributions of alleles of the 3 polymorphisms in the D2d study were similar to those reported in the UK Biobank of participants with prediabetes.5 Consistent with the UK Biobank study and other studies,5,16,17 the 25(OH)D levels achieved during the D2d trial did not differ significantly among participants with different VDR polymorphisms.
In the UK Biobank study, among adults with prediabetes and a median 25(OH)D level of 19.2 ng/mL (a value below our referent range of 20–29.9 ng/mL), there was a stepwise decrease in the risk of diabetes at 25(OH)D levels of lower than 10 (the study’s referent), 10 to 20, 20 to 30, and 30 ng/mL or higher.5 Risk reduction was present in all VDR genotypes of the 4 examined polymorphisms (ApaI, BsmI, TaqI, and FokI), but it was more prominent among those carrying the T allele of BsmI. There were too few participants in the D2d study with sufficiently low 25(OH)D levels to address this range of the 25(OH)D spectrum. Conversely, there were too few participants with sufficiently high 25(OH)D levels in the UK Biobank study to address the question posed in our study. To our knowledge, no other high-dose vitamin D trials among adults with prediabetes have examined how VDR polymorphisms may modify the effect of vitamin D supplementation on diabetes risk.
Our exploratory findings, if confirmed, hold promise for high-dose vitamin D3 as a targeted, personalized approach to reducing the risk of type 2 diabetes among selected adults with prediabetes. The magnitude of the observed risk reduction among participants with AC and CC alleles of the ApaI polymorphism, if confirmed in an independent clinical trial, would have clinical implications for the management of prediabetes. In the original report of the D2d trial,2 the HR for conversion to type 2 diabetes with vitamin D supplementation was 0.88 (95% CI, 0.72−1.04). The HR decreased to 0.81 (95% CI, 0.66−0.99) in our exploratory analysis when genetically nonresponsive participants (those with AA alleles of the ApaI polymorphism, comprising 29.5% of all participants) were excluded. If confirmed, a 19% risk reduction in conversion to type 2 diabetes with vitamin D3 supplementation would not be trivial. First, assessment of a single VDR polymorphism is inexpensive and now widely available.