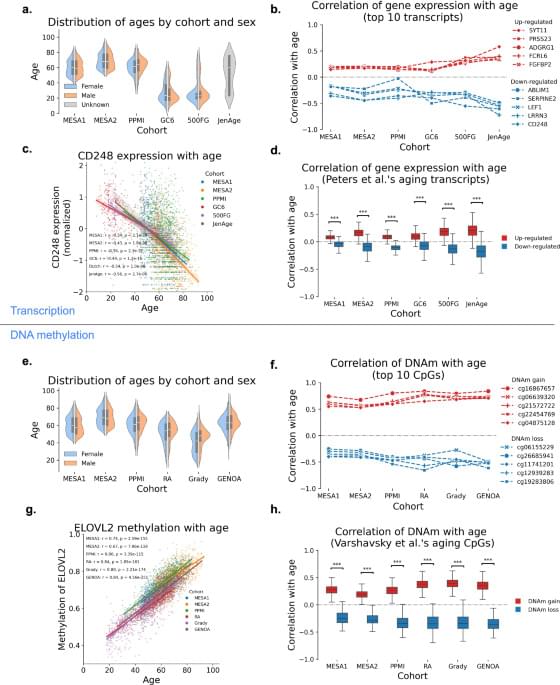
DNA methylation and gene expression data integration identify aging-related genes in blood that predict health outcomes, offering new insights into aging biology and potential therapeutic targets.

Artificial intelligence is rapidly advancing to the point where it may be able to write its own code, potentially leading to significant job displacement, societal problems, and concerns about unregulated use in areas like warfare.
## Questions to inspire discussion.
Career Adaptation.
🎯 Q: How should workers prepare for AI’s impact on employment? A: 20% of jobs including coders, medical, consulting, finance, and accounting roles will be affected in the next 5 years, requiring workers to actively learn and use large language models to enhance productivity or risk being left behind in the competitive landscape.
Economic Policy.
📊 Q: What systemic response is needed for AI-driven job displacement? A: Government planning is essential to manage massive economic transitions and job losses as AI’s exponential growth reaches a tipping point, extending beyond manufacturing into white-collar professions across multiple sectors.

A positive newborn screening for spinal muscular atrophy (SMA) is currently considered a medical emergency. Without early treatment, severe disability or death in infancy are likely. However, research findings from Germany and Australia now show that in rare cases, a positive screening result can be a genetic false alarm. Researchers have discovered that functional tests in a zebrafish model may enable fast and reliable clinical decision-making in cases of unclear genetic findings.
The study “SMN1 variants identified by false positive SMA newborn screening tests: Therapeutic hurdles, and functional and epidemiological solutions” was published in the American Journal of Human Genetics and another study, “Clinical relevance of zebrafish for gene variants testing. Proof-of-principle with SMN1/SMA,” in EMBO Molecular Medicine. The collaborative research team was led by Professor Dr. Brunhilde Wirth, Director of the University of Cologne’s Institute of Human Genetics and Principal Investigator at the Center for Molecular Medicine Cologne (CMMC) and Dr. Jean Giacomotto from Griffith University’s Institute for Biomedicine and Glycomics, Brisbane, Australia.
The scientists examined two newborns—a girl from Germany and a boy from Australia—in whom routine screening initially failed to detect the SMN1 gene. A missing SMN1 gene is the main genetic trigger of SMA. This diagnosis would normally result in immediate treatment, as it would be assumed that the child’s life is in danger. However, further genetic analysis revealed a surprising finding: both children carried rare SMN1 variants that had not been detected by the screening test. It remains unclear whether these variants cause the disease.
Scientists have traditionally studied how the brain controls movement by asking patients to perform structured tasks while connected to multiple sensors in a lab. While these studies have provided important insights, these experiments do not fully capture how the brain functions during everyday activities, be it walking to the kitchen for a snack or strolling through a park.
For people living with Parkinson’s disease, this gap between laboratory research and real-world behavior has limited efforts to improve gait symptoms outside of the clinic.
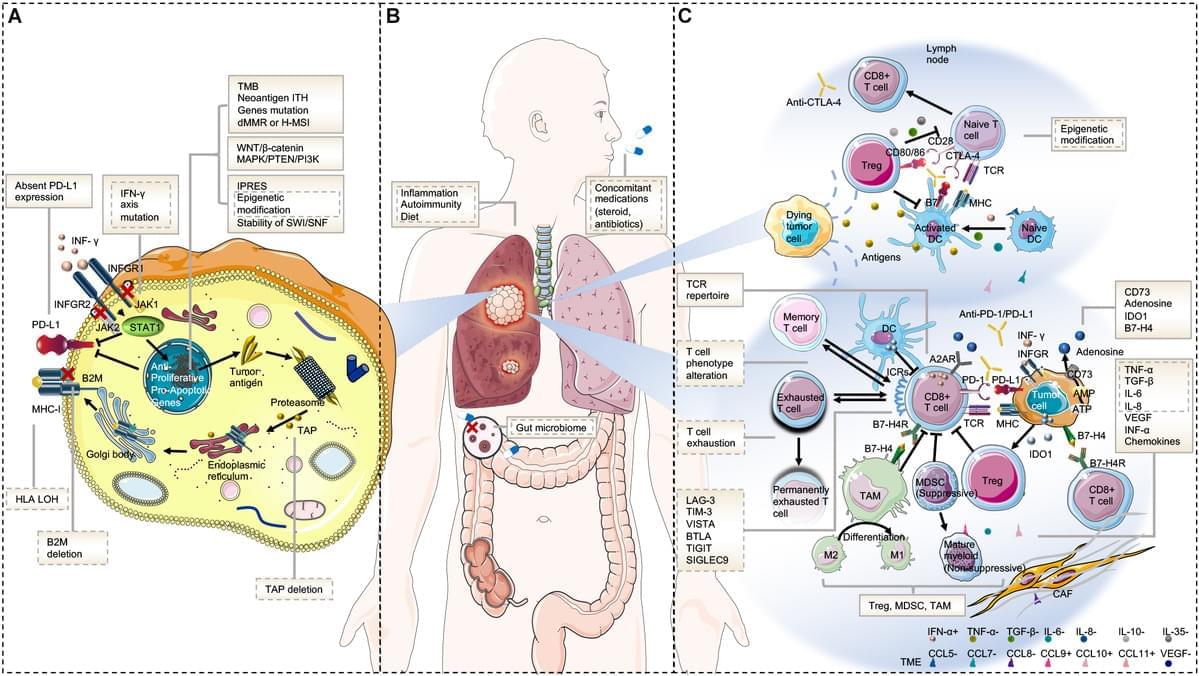
Immunotherapy has revolutionized lung cancer treatment in the past decade. By reactivating the host’s immune system, immunotherapy significantly prolongs survival in some advanced lung cancer patients. However, resistance to immunotherapy is frequent, which manifests as a lack of initial response or clinical benefit to therapy (primary resistance) or tumor progression after the initial period of response (acquired resistance). Overcoming immunotherapy resistance is challenging owing to the complex and dynamic interplay among malignant cells and the defense system. This review aims to discuss the mechanisms that drive immunotherapy resistance and the innovative strategies implemented to overcome it in lung cancer.
The discovery of the immune checkpoint inhibitors (ICIs), represented by the monoclonal antibodies that block cytotoxic T−lymphocyte−associated protein 4 (CTLA-4), programmed death protein 1 (PD-1), and programmed death protein ligand 1 (PD-L1), has revolutionized the therapeutic landscape of lung cancer. The significant survival benefit derived from ICI-containing treatment has established it as the mainstay first-line therapy in patients with advanced or locally advanced non-small cell lung cancer (NSCLC) and extensive small-cell lung cancer (SCLC). Unprecedented long-term clinical benefit or even, in some cases, a complete recovery has been witnessed in lung cancer, particularly in patients with high PD-L1-expressing tumors (1– 3). Currently, investigations are under way aimed at integrating immunotherapy in the treatment of early-stage lung cancer.
However, most patients with NSCLC develop primary resistance during ICI monotherapy and only 15 to 20% achieve partial or complete response (3). Acquired resistance also occurs in initially responding patients with advanced NSCLC treated with ICIs, after a median progression-free survival (PFS) of 4–10 months (4– 9). The mechanisms of resistance to immunotherapy are not yet fully understood, and methods to overcome them must be developed. Herein, we discuss the pathways driving resistance to immunotherapy in lung cancer to help clinicians in their current practice, as well as identify future research priorities and treatment strategies.
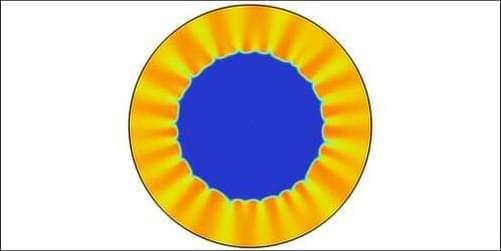
For a phenomenon like a wildfire burning through a forest or a disease moving through a population, the resulting patterns can sometimes be modeled using a so-called reaction–diffusion system—an experiment where a chemical reaction front moves through a region full of reactants. Now Anne De Wit of the Université Libre de Bruxelles and her colleagues have demonstrated that new patterns can be revealed when the reactants flow against the direction of the front’s propagation, causing it to freeze in place [1]. Their “sun-ray” pattern is the first one discovered this way, but the technique could generate other patterns that might replicate behavior in forest fires or epidemics.
Two years ago, De Wit and her colleagues brought a propagating reaction front to a standstill by slowly and continually injecting a reactant into the center of a disk-shaped chamber filled with the other reactant, against the front’s inward propagation [2]. The stopping occurred when the outward flow matched the rate at which the inner reactant was consumed. De Wit says that a stationary front allows more control and thus more careful study of patterns than a propagating front.
As a demonstration of this control, the researchers have now used reactants with different diffusion rates in the same outward-flow setup. In this case, the stationary front developed ripples—an effect previously seen in propagating fronts. The researchers also observed radial “rays”—narrow regions of higher concentration of one of the reactants. They showed in simulations and experiments that properties of the front can be precisely controlled by varying the flow rate.
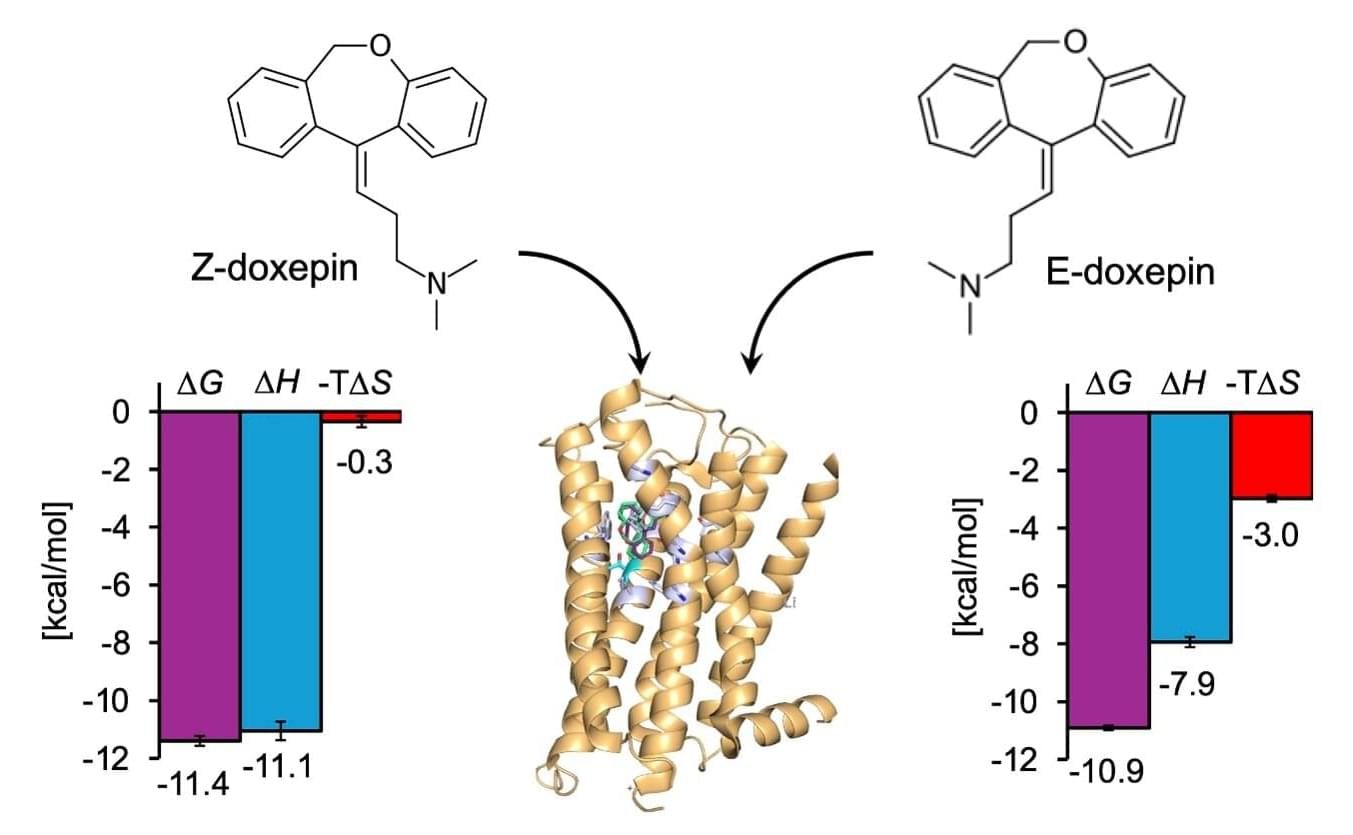
G-protein-coupled receptors (GPCRs) are one of the largest families of cell surface proteins in the human body that recognize hormones, neurotransmitters, and drugs. These receptors regulate a wide range of physiological processes and are the targets of more than 30% of currently marketed drugs. The histamine H1 receptor (H1R) is one such GPCR subtype that plays a key role in mediating allergic reactions, inflammation, vascular permeability, airway constriction, wakefulness, and cognitive functions in the human body. While antihistamines primarily target H1R, current drugs can exhibit limited therapeutic efficacy, prompting researchers to look at H1R ligands from new perspectives.
Recently, the importance of drug design based not only on the affinity or binding energy between a compound and its target protein, but also on its components, enthalpy, and entropy, has been recognized as crucial for rational drug design. In particular, enthalpy–entropy compensation has emerged as a key concept for understanding ligand selectivity and isomer specificity. However, direct experimental measurement of these thermodynamic parameters has been limited to cell surface proteins, such as GPCRs.
Addressing this gap, a research team led by Professor Mitsunori Shiroishi from the Department of Life System Engineering, Tokyo University of Science (TUS), Japan, systematically investigated the binding thermodynamics of the H1R. The team included Mr. Hiroto Kaneko (first-year doctoral student) and Associate Professor Tadashi Ando from TUS, among others. Their study was published online in ACS Medicinal Chemistry Letters on January 26, 2026.

Humans develop sharp vision during early fetal development thanks to an interplay between a vitamin A derivative and thyroid hormones in the retina, Johns Hopkins University scientists have found. The findings could upend decades of conventional understanding of how the eye grows light-sensing cells and could inform new research into treatments for macular degeneration, glaucoma, and other age-related vision disorders. Details of the study, which used lab-grown retinal tissue, are published today in Proceedings of the National Academy of Sciences.
“This is a key step toward understanding the inner workings of the center of the retina, a critical part of the eye and the first to fail in people with macular degeneration,” said Robert J. Johnston Jr., an associate professor of biology at Johns Hopkins who led the research. “By better understanding this region and developing organoids that mimic its function, we hope to one day grow and transplant these tissues to restore vision.”

This multicenter case series highlights 5 cases of enterovirus encephalitis among people with MS receiving ocrelizumab, presenting with fever, encephalopathy, and gait changes, as well as myocarditis in 1 case.
ObjectivesAnti-CD20 therapies for multiple sclerosis (MS) are highly effective at preventing disease activity. Recognizing infectious complications of these therapies is essential. MethodsThree MS centers shared deidentified clinical data on persons with MS (pwMS) receiving ocrelizumab who developed enterovirus encephalitis.
Even before a tumor in the pancreas becomes discernible, an activated cancer gene actively remodels its future environment and creates an inflammatory and immune-defensive microenvironment in which the carcinoma can grow. This has been shown by an international research team led by Ulm University in a pioneering study. The scientists’ study opens up new possibilities for developing personalized intervention strategies—before a difficult-to-treat tumor even develops.
It is one of the most aggressive forms of cancer: Pancreatic cancer is usually diagnosed late because it initially causes no symptoms and therefore goes unnoticed. In addition, it is highly metastasizing. Once pancreatic cancer is finally identified, a cure is often no longer possible.
A research team from the Institute of Molecular Oncology and Stem Cell Biology (IMOS) at Ulm University, together with national and international partners, has made a ground-breaking discovery that could pave the way for a much earlier diagnosis: The oncogene KRAS —the main driver of pancreatic cancer—creates its own environment, providing best growth conditions for the carcinoma and in which immune defense T-cells cannot penetrate. The results of the study have now been published in the journal Molecular Cancer.