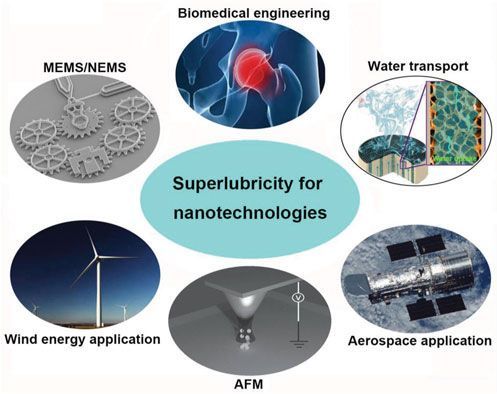
Achieving near-zero friction in commercial and industrial applications will be game-changing from tiny microelectromechanical systems that will never wear out, to oil-free bearings in industrial equipment, to much more efficient engines and giant wind turbines scavenging energy even in low wind conditions. Superlubricity offers promising solutions to overcome lubrication challenges in various areas of nanotechnology including micro/nano-electromechanical systems (MEMS/NEMS), water transport control, biomedical engineering, atomic force microscopy (AFM), aerospace and wind energy applications, as well as other electronic devices. It is one of the most promising properties of functional nanomaterials for energy saving applications.