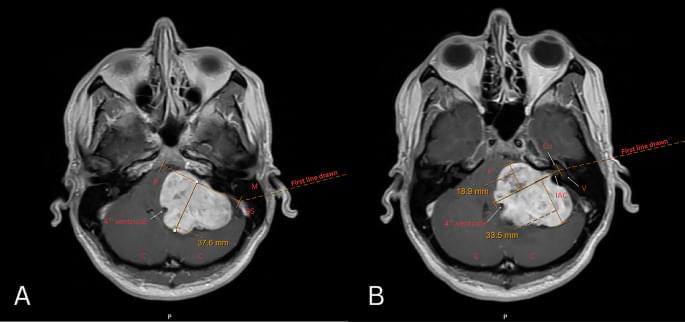
While many American adults are trying to reduce cholesterol levels, certain cancerous tumors have a relentless appetite for the metabolite. Some tumor cells use as much cholesterol as they can access to accelerate their growth beyond the capabilities of normal cells.
Turning tumors’ cholesterol cravings into weakness Scientists at Sanford Burnham Prebys Medical Discovery Institute and their collaborators at the University of Illinois Chicago have published findings in Science Advances regarding a potential method for turning the tables on these tumors by subverting their cholesterol cravings. The researchers revealed new insights into enzymes that help move cholesterol around cells. Without the help of these enzymes, a cholesterol traffic jam occurs, blocking the cancer cell’s ability to fuel tumor growth.
Cancer cells with a mutation in the tumor-suppressing TP53 gene are known to produce extra cholesterol. This may make them more vulnerable to starvation if scientists can put a stop to the steady supply of the lipid.