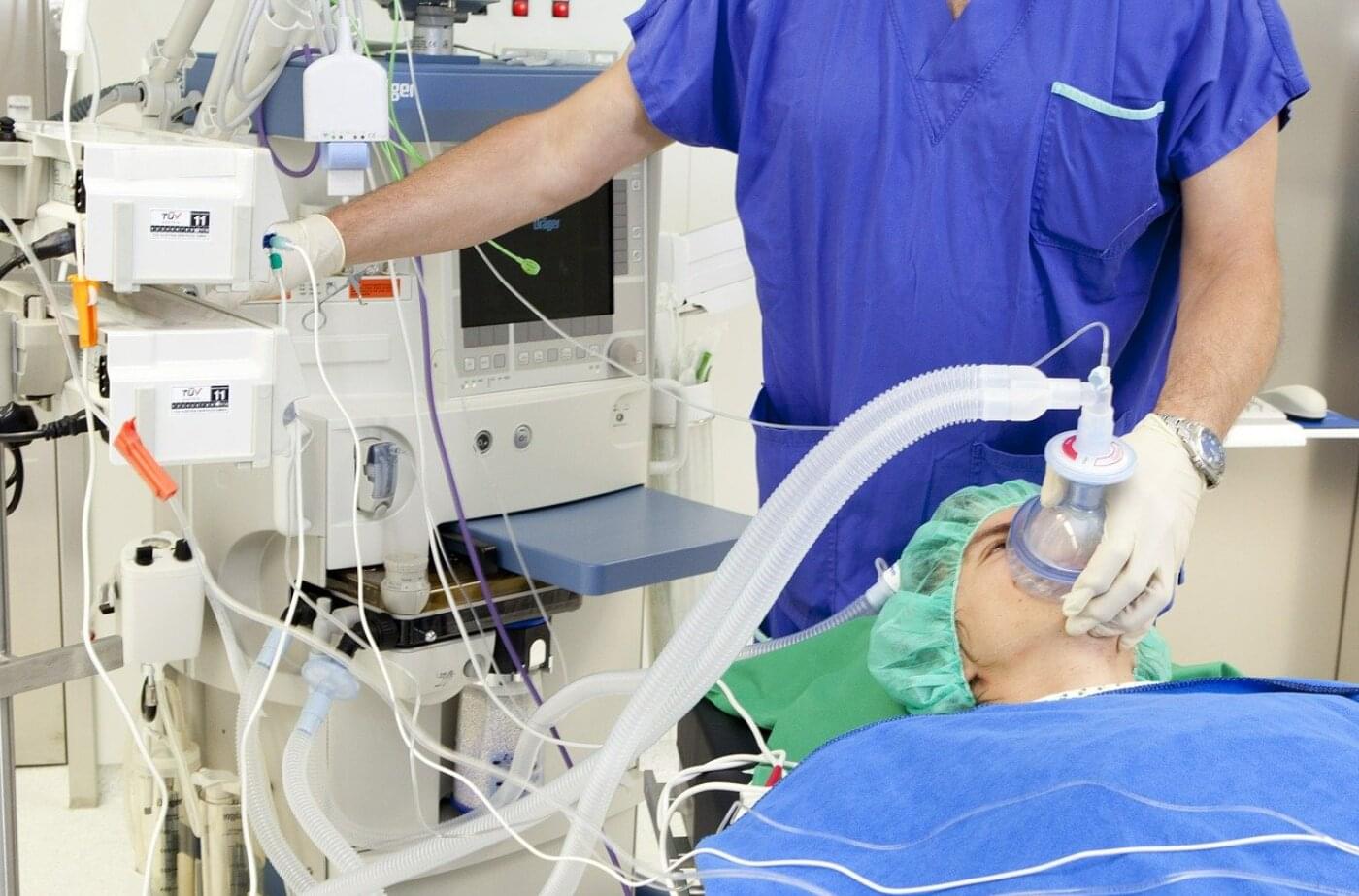
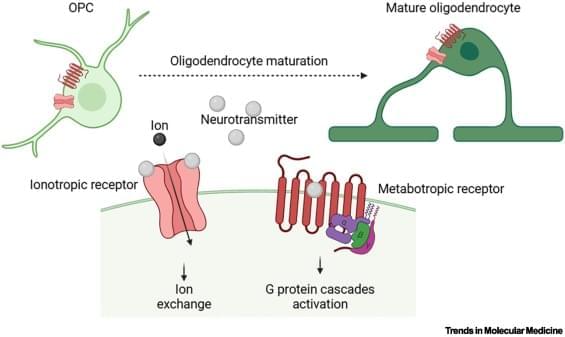
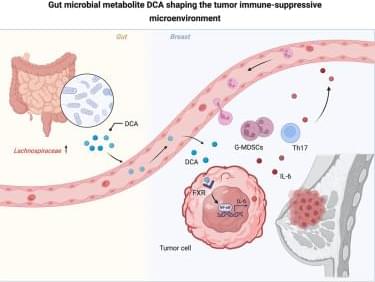
People often describe anesthesia as something that puts a patient in a “deep sleep.” An anesthesiologist enters the operating room, and part of their mission is to ensure that the patient is completely unaware of what is happening around them until they wake up, often several hours later. Scientists and doctors have long debated what happens to the brain under anesthetic drugs during a surgical procedure.
A new study by Yale School of Medicine’s Departments of Anesthesiology and Neurology published on May 11, 2026, in Proceedings of the National Academy of Sciences uncovers new insights which may change the way we describe being under anesthesia. The study, “Spectral mapping reveals a resemblance of the anesthetic brain state to both sleep and coma,” reveals that being anesthetized may be more than simply being “put to sleep.” It can potentially carry more similarities to being in a coma than we originally thought.