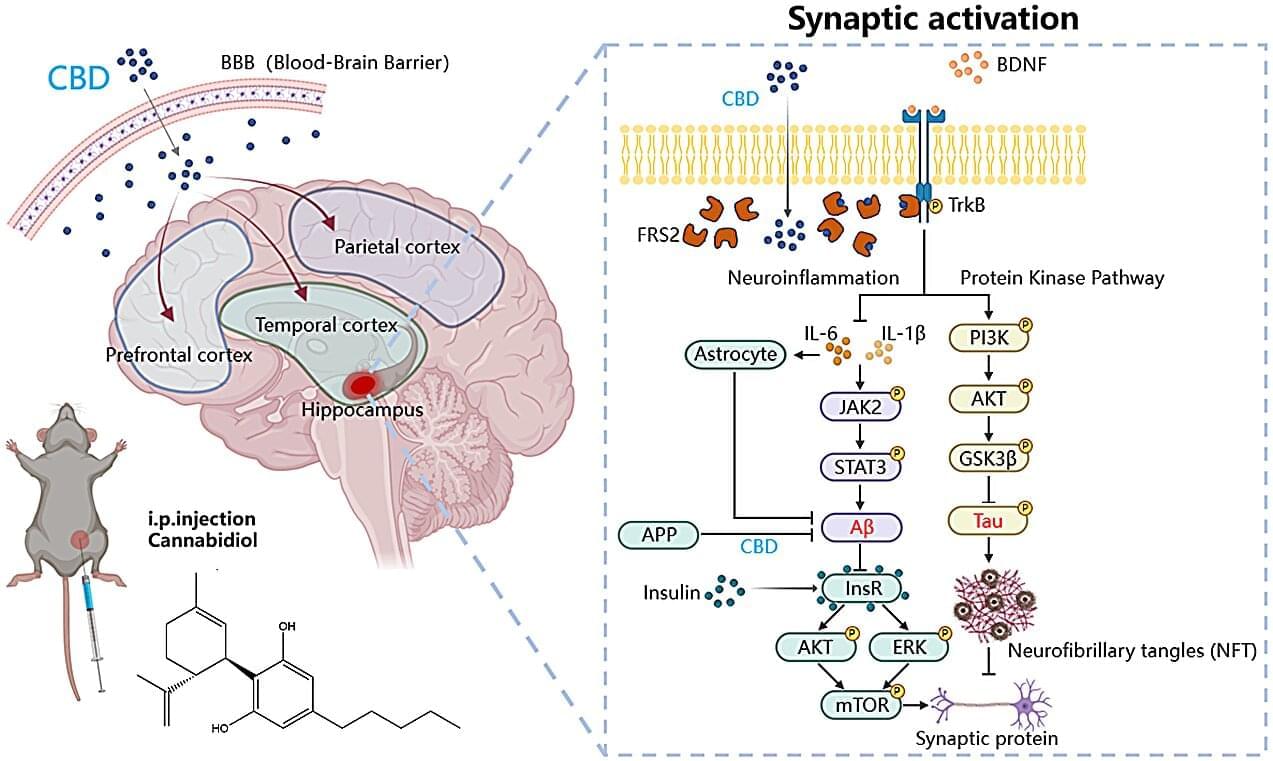
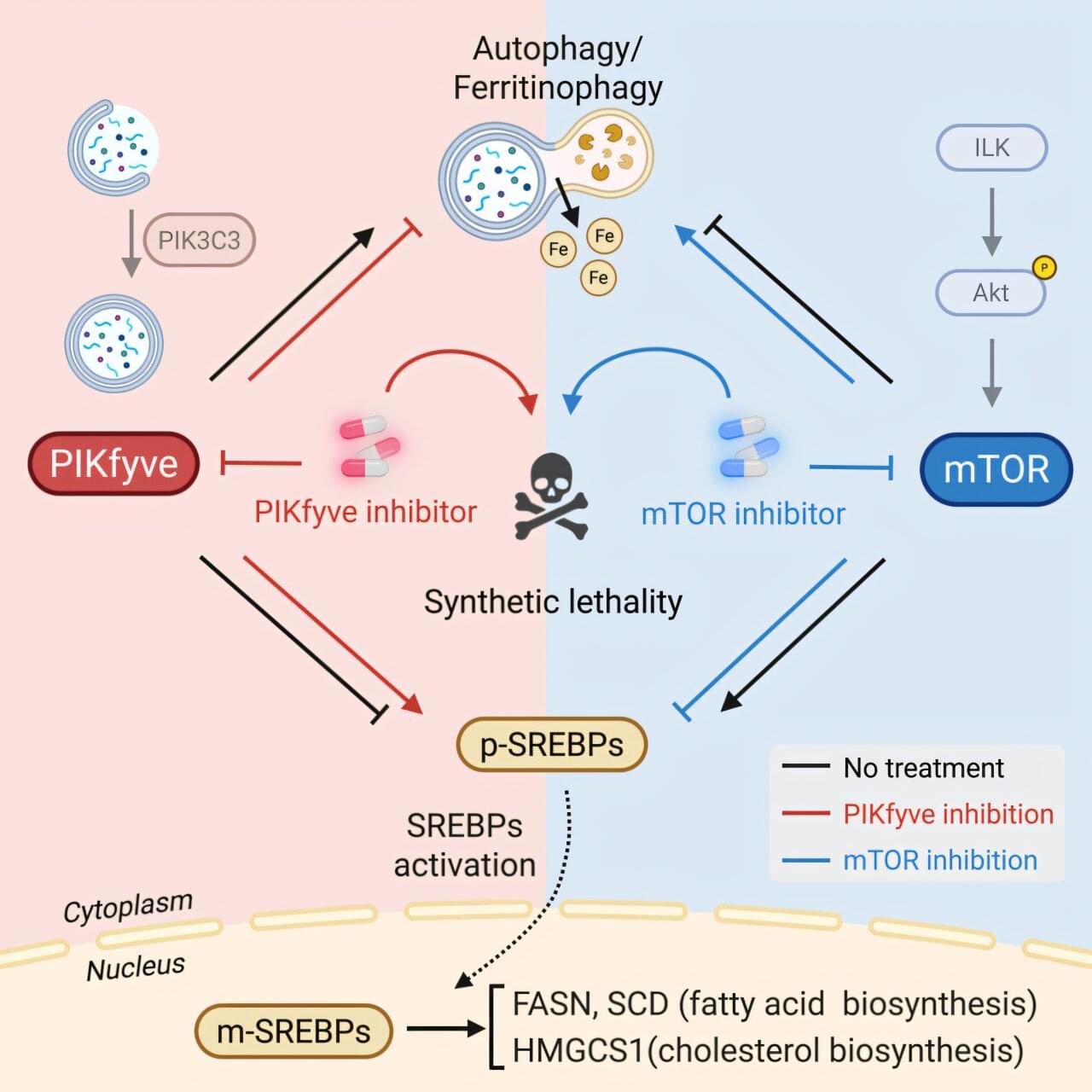
Repairing or degrading damaged lysosomes.
Many key lysosomal repair proteins have an intrinsic propensity to form biomolecular condensates. This points to an emerging paradigm where phase separation, not just individual protein actions, may be a central feature in orchestrating the response to membrane damage.
Recent work has separately highlighted the roles of protein condensates and lipid domains in membrane repair. This raises the intriguing possibility of a mechanistic synergy, where protein and lipid phase separation coregulate each other to mount an integrated response to damage.
A key question is how cells choose between the repair and degradation of a damaged lysosome. The recent discovery of pathways that sense lipid packing defects suggests a new framework, where the biophysical state of the membrane itself helps determine organelle fate. sciencenewshighlights ScienceMission https://sciencemission.com/lysosome-integrity
Lysosomes are sophisticated signaling hubs whose function depends on membrane integrity. A breach of this barrier, known as lysosomal membrane permeabilization, triggers inflammation and cell death, driving pathologies from lysosomal storage disorders to neurodegeneration. Cells counter membrane damage with diverse repair mechanisms, including endosomal sorting complexes required for transport machinery, sphingomyelin scrambling, annexin-mediated scaffolding, lipid transport, and stress granule plugging. This diversity suggests singular strategies are insufficient, posing an ‘orchestration challenge’ regarding precise initiation, spatial organization, and temporal coordination. This opinion article proposes that biomolecular condensation, initiated by damage cues, acts as a primary organizing principle.