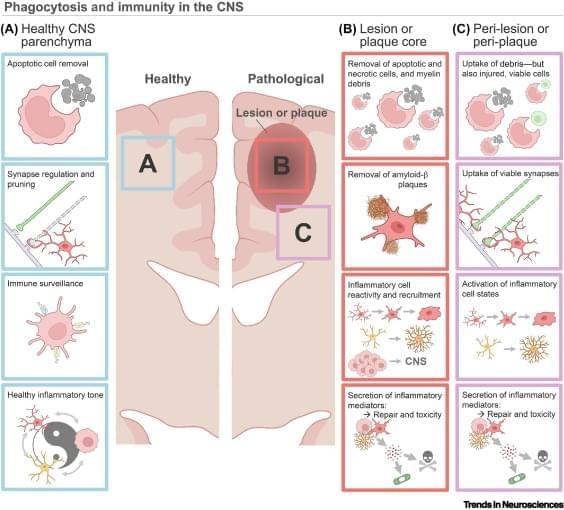
Brain phagocytosis and neuroinflammation.
Phagocytes in the central nervous system (CNS), including astrocytes, microglia, and macrophages, shape development and homeostasis by pruning synapses and removing apoptotic debris.
Phagocytosis is mediated by various ligand–receptor dyads and signaling pathways, enabling CNS phagocytes to respond to neuroimmune shifts across the lifespan and during pathology.
Phagocytosis pathways regulate recovery in various models of CNS pathology, including multiple sclerosis, CNS injury, ischemic stroke, and age-associated neurodegeneration.
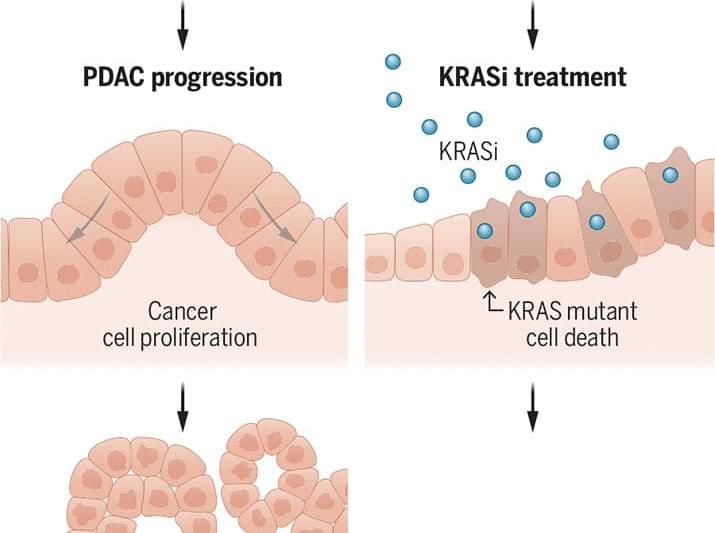
Phagocytosis pathways are intimately integrated with the inflammatory cell state and remove viable cells in pathology-adjacent tissue, highlighting the complexity of targeting these systems.
To maximize benefit and minimize off target damage, new phagocytic-based approaches should optimize drug delivery timing and location, tailored to each CNS pathology. sciencenewshighlights ScienceMission https://sciencemission.com/resolution-of-inflammation